Highly Ordered Amorphous Silion-Carbon
Alloys Obtained by RF PECVD
I. Pereyra, C. A. Villaorta,M.N.P. Carre~no,
Depto. deEng. Sistemas Eletr^onios,EsolaPolitenia,
UniversidadedeS~aoPaulo,S~aoPaulo,Brasil
R.J. Prado,and M.C.A. Fantini
Institutode Fsia, UniversidadedeS~aoPaulo,S~aoPaulo,Brasil
Reeived15February,2000;reeivedinnalformon29Marh,2000
WehaveshownthatlosetostoihiometryRFPECVDamorphoussilionarbonalloysdeposited
undersilanestarvingplasmaonditionsexhibitatendenytowards-SiChemialorder. Motivated
bythis trend,wefurtherexploretheeetofinreasingRFpowerand H2 dilutionofthegaseous
mixtures, aiming to obtain the amorphous ounterpart of -SiC by the RF-PECVD tehnique.
Doping experimentswere also performed on ordered material using phosphorus and nitrogen as
donorimpuritiesandboronandaluminumasaeptorones.FornitrogenadopingeÆienyloseto
deviequalitya-Si:Hwasobtained,thelowerativationenergybeing0,12eVwithroomtemperature
darkondutivityof2.10 3
(.m). NitrogendopingeÆienywashigherthanphosphorousforall
studiedsamples. For p-typedoping, results indiatethat, eventhough theattainedondutivity
valuesare not devielevels, aluminumdopingondutedto a promisingshift inthe Fermi level.
Also, aluminum resulted a more eÆient aeptor than boron, in aordane to observations in
rystallineSiCmaterial.
I Introdution
Thedepositionofamorphoushydrogenatedsilion
ar-bide thin lms,a-Si
1 x C
x
:H, byPECVDhasbeen
ex-tensivelystudiedduetotheabilityofthistehniqueto
ontrolthearbon ontentin thelms and, therefore,
the optialgap (E
o
). Theinreasein the optialgap,
following the inrease in the arbon onentration, is
desirable formany devie strategiesand hasbeenone
of themain appeals of thesealloys. However,forhigh
arbonontentlms(x0:5), aderease in the
opti-algapisgenerallyobserved,whihleadstoadiÆulty
in obtaining a material with E
o
> 2;5 3 e V [1℄.
This derease in the optial gap observed for arbon
rih a-Si
1 x C
x
:H has been related to the existene of
graphite-likesp 2
C-Cbondinginsteadof diamond-like
sp 3
C-Cbonding[1,2℄.
Solomonando-workers[3℄showedthat in thelow
powerregime,wheretheRFpowerissuÆienttobreak
theSiH
4
moleulesbutnottheCH
4
ones,thepresene
of graphite-likebonds is avoided. Howeverthearbon
to silion relative onentration in the solid phase is
limitedupto 50%andtheresultingmaterialisrihin
methylated radials. Reently, we have demonstrated
thatitispossibletoobtainnonmethylatedPECVD
a-SiC:Hthinlms(fromSiH +CH )witharbonto
sili-onratiohigherthan0.5(upto0.7)withC-Cbonds
o-urringmainlyinthesp 3
(diamond-like)hybridization,
optialgapashighas3.7eVandhigheletrial
resistiv-ity[4-6℄. Theresultsdemonstratethatthese
diamond-likesilionarbidelmsareobtainedinaspeial
depo-sitionondition,alled\silanestarvingplasma"regime
[6℄. This regime is a partiular aseof the low power
density one where, for a ritially low SiH
4
ow, the
lm growth is ontrolled by the spare SiH
4
radials,
simplifying the plasma hemistry and promoting
ar-boninorporationinthesp 3
hybridization. Inthisway
wehaveshownthatthestarvingplasmadeposition
on-ditionshavearuialroleinthegrowthofloseto
sto-ihiometrymaterialexhibiting atendeny towardsthe
hemial order of rystalline silion arbide,this is to
say eah silion atom (tetrahedrally) bonded to four
arbon atoms and eah arbon atom bonded to four
silionatoms. Forarbonrihsamplesthis deposition
regimepromotestheprodutionoflmswithveryhigh
valuesoftheoptialgap,stronglysuggestingthe
inor-porationofarbonin sp 3
bonds.[1℄
Chemial order in silion arbon alloys has been
a ontroversial point. Cardona et. al. in their
pio-neeringwork[7℄,andmanyauthorsafterthem,argued
thatsilionandarbonatomsarerandomlydistributed,
the other hand, Evangelisti et al.[8℄ have reported a
strongtendenytowardsthehemialorderof-SiCin
a-Si
1 x C
x
:H lms. Our work indiates that material
grown in the low powerregime out of silane starving
onditions are silion rih randomly distributed while
the material grown with silane starving plasma
on-ditions is lose to stoihiometry or arbon rih and
exhibits a preferene for hetero-nulear bonding (SiC
hemialorder).
Enouragedby the hemial order exhibited by
a-Si
1 x C
x
:Halloysproduedinthesilanestarvingregime
we further explore the possibility of inreasing even
morethe hemial order to promote thegrowth of an
amorphousounterpartof-SiCorevenSiC
mirorys-tals (-SiC).Wealsoanalyzethenand ptypedoping
eÆienyofthese a-Si
1 x C
x
:Hlms.
Itisknownthathydrogenethingduringthegrowth
of amorphous silion has a very important eet on
thestrutural properties ofa-Si:H lms[9℄andH
2
di-lution of the gas mixture is a tehnique widely used
to produe mirorystalline Si. Inthis way, some
au-thorshaveusedhydrogendilutionandhigherRFpower
densityto produemirorystalline p-type silion
ar-bon (boron doped) alloys. In fat they obtained
sili-onmiro-rystalsembeddedinanhydrogenated
amor-phous silion arbon alloy network [10,11℄. At the
present time, to our knowledge, there is no report of
SiCmiro-rystalsobtainedbyRF-PECVDwhih
jus-tiesourinterestinpursuingthistasktakingadvantage
ofthepromising propertiesof thea-SiC:Hobtainedin
silanestarvingplasmaonditions.
Similarly, thestruturalorder in a-SiC:H grown in
\silane starving plasma" regime is a very important
harateristifordopingexperiments,sineitisan
uni-versalfat thatamorphoussilionbasedalloysexhibit
anenhaneddegreeofdisorderforinreasingalloy
ele-mentontent,whihdereasesthearriermobilityand
preventstheourreneofaneÆientdoping, limiting
theirdevie appliations. Inthiswaywestudy the
in-ueneofthehemialorder,induedbythe\starving
plasma"onditiononthen-typeandp-typedoping
ef-ienyofPECVDa-Si
1 x C
x :H.
II Experimental details
a-Si
1 x C
x
:Hlmswerepreparedbytheradiofrequeny
(RF) Plasma Enhaned Chemial Vapor Deposition
(PECVD) tehnique, with an RF frequeny of 13,56
MHz, from appropriated gaseous mixtures of SiH
4 + CH 4 + H 2
in a apaitively oupled reator. A more
detailed desription of the deposition system an be
foundelsewhere[12℄. RFpowerdensity, methane
on-entration, and hydrogen dilutionof the gaseous
mix-tures were varied. Substrate temperature for all the
sampleswas300 o
Candsilaneowwaskeptatthe
min-regime".
To establish the role of the \starving plasma" in
the doping eÆieny of a-SiC:H, therst experiments
were arried outto ompare the performane of
sam-ples grownin and outof this ondition. Forthis
pur-pose, both types of samples were ion-implanted with
310 20
m 3
phosphorous and nitrogen. More details
ontheion-implantationarereportedelsewhere[13℄.
Based on the results obtained for samples grown
withH
2
dilutionofthegaseousmixtureandonn-type
doping, in ase of the p-type doping experiments we
entered onsamplesgrownin\silanestarvingplasma"
with H
2
dilution. Sinegoodresultsin rystallineSiC
have been obtained with aluminum, we utilized this
dopingelementasaeptorimpurity. Howeverthis
im-purity had to be introdued in the lms throughlow
temperaturethermaldiusionexperimentsdueto
teh-nial diÆulties with the aluminum ion implantation.
Aluminumdiusionwasarriedoutbythermally
evap-orating a thin aluminum layer followed by a thermal
annealingstep. Theanalyzedannealingstepswere350
o
Cfor4,8and12hours and450 o
Cfor 0,5,2, 4and
6hours.
Thelmsstrutureandbondingwereharaterized
byFouriertransforminfrared(FTIR)spetrosopy,
op-tial absorption and extended X-ray absorption ne
struture (EXAFS). The atomi onentrations of Si,
C, N and P were determined by Rutherford
bak-sattering (RBS) using a 2.3 MeV He +
beam, with
satteringangleof170 o
. TheHontentwasdetermined
throughforwardreoilspetrometry(FRS)witha
tar-gettiltangleof75 o
and30 o
detetionangle. The
trans-portpropertiesof doped sampleswere studied
analyz-ing the thermal dependene of the dark ondutivity,
d
(T), measured in a vauum ryostat. The thermal
ativationenergy(E
a
)wasdeterminedfromthe
Arrhe-nius plot. In those plots the data were tted with a
funtion ln[
d
(T)℄ =A+B=T and the ativation
en-ergy wasobtained from the slopesof those funtions.
Thereproduibilityoftheseresultsleadtoanestimated
errorless than10%for theondutivity. A 10%error
for the ativation energy wasobtained byomparison
ofthemaximumandminimumslopesintheArrhenius
plot.
III Results and disussion
III.1. H
2
Dilution and RF power
Fig.1showstheoptialgapE
o
asafuntionofCH
4
onentrationforsamplesgrownunderstarvingplasma
onditionswith andwithouthydrogendilutedgaseous
mixtures. It is observedthat for samplesgrown
with-out H
2
dilution the optial band gap inreases slowly
forCH
4
onentrationvaluesupto around77%,while
H
2
dilution show a similar but more pronouned
be-havior,thetransition ourringathighervaluesofthe
CH
4
onentration(approximately88%). This
transi-tion is assoiated with the passage over the
stoihio-metri omposition (x =0.5), sothese resultssuggest
alowerarboninorporationin thesolidphasefor
hy-drogendiluteddepositedmaterial.
Figure 1. Optial gap as afuntion of the methane
on-entration forsamples depositedat aRFpowerdensityof
25mW/m 2
andwithaH2owof100sm()andwithout
H2 dilution(Æ).
InFig.2theintegratedabsorptionoftheIR
absorp-tionpeakat780m 1
,assignedtotheSi-Cstrething
vibrations[1℄,dividedbythesamplethiknessas
fun-tion of methane volume fration is depited, for the
samesamplesinFig. 1. Amaximumin theintegrated
absorption is observedforbothsets of samples.
How-ever the peak position is shifted towards higher CH
4
onentrations for the H
2
diluted samples. Atually
this maximumis anindiationthat the stoihiometri
onentrationx=0.5hasbeenpassedthrough,sinefor
higherarbonontentthedensityofSi-Cbondsshould
derease and the C-C bonds should inrease. In this
waythis result indiates,in aordanewith the RBS
measurements and the optial gap results, that
stoi-hiometry is attained for higher CH
4
onentrations
for samples deposited with H
2
diluted gas mixtures.
Forsamplespreparedat 25mW/m 2
andwith H
2
di-lution no traes,within thedetetion limitof 1% of
the FTIRtehnique,ofCH
n
bonds areobserved. The
onentrationofSiH
n
isalsolowerthanin lmsgrown
withoutH
2
dilutionandbeomesverysmallforaCH
4
onentrationof98%. ThismeansthatforinreasingC
ontentinthesolidphase,thepolymeriphasealways
presentin thehydrogenatedamorphoussilionarbon
alloyshasbeenalmosteliminatedbythe\ething
pro-ess"andverylowhydrogenontentisobtained.
Havingidentiedthemethaneonentrationleading
to losetostoihiometrymaterialforhydrogendiluted
gaseousmixtures and aRFpowerof 25 mW/m 2
, we
hosethisonditiontoperformourstudiesontheeet
ofinreasingH
2
dilutionandRFpowerdensitiesonthe
strutural propertiesofthelms.
Figure2. IntegratedabsorptionintensityfortheSi-Cbond
vibrationpeak vs. methane onentration for samples
de-positedataRFpowerdensityof25mW/m 2
withaH
2 ow
of100sm()andwithoutH2dilution(Æ).
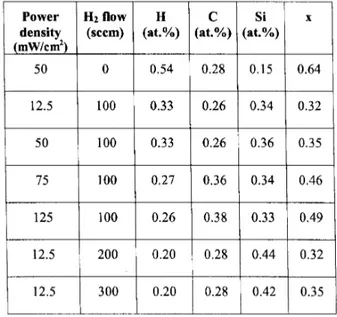
In Table1 the RBS results are summarized. It is
observed that inreasing RF power inreases the
ar-bonontentanddereasesthehydrogenonentration
ofthelms,inreasinghydrogenowdereasedthe
hy-drogenontentevenfurther.
Table1: Compositionofthestudiedsamplesobtained
fromRBSmeasurements.
TheFTIRspetraforsamplesgrownwithdierent
H
2
ow, depitedin Fig. 3showanenhaned
absorp-tion band at 780 m 1
. Sine the sample deposited
without hydrogen dilution and 90% methane volume
onentration is arbon rih as indiated by the RBS
measurements[14℄theinreaseintheSi-Cbandanbe
attributedtothefatthatthesamplesaregettingloser
attakspreferentiallyarbonbonds. Thishypothesisis
onrmedbyRBSmeasurements,whih showthatthe
Si/Cratioinreasesforinreasing H
2
dilution. Atthe
sametime, the CH
n
and theSi-CH
n
strething
vibra-tionabsorptionbands(inthe2800m 1
to3000m 1
and 1250 to 1350 m 1
spetral regions, respetively)
dereaseandforH
2
owhigherthan100smandthe
CH
n
related bands disappear whih is also onsistent
withthehypothesisofpreferentialarbonething. The
FTIRspetraalsoshowapronouneddereaseintheH
ontentofthelmsforinreasingH
2
ow,revealed by
theCH
n
andSiH
n
bondingongurations.Itisalso
ob-servedthattheSi-Hstrethingabsorptionbandloated
at 2090 m 1
, dereases but it does not disappear as
theCH
n
bands,whihisonsistentwiththehypothesis
ofpreferentialethingoftheC-Hbonds.
Figure3. InfraredspetraforsamplesdepositedwithaRF
powerdensityof25mW/m 2
,90%methanevolumefration
andvaryingH
2 ow.
Inorder tostudytheeet ofinreasingRFpower
densitiesasampleset,varyingtheRFpowerdensity
be-tween12,5and 500mW/m 2
andkeepingonstantall
theotherdepositionparameters,wasprepared. Itwas
alsofoundthat,astheRFpowerdensityinreases,the
depositionrateinreases. Thisresultindiatesthatfor
higher RF power the deposition mehanism inreases
more rapidly than the ething one. In Fig. 4 an
in-rease in the Si-C absorption band for inreasing RF
powerdensities,upto250mW/m 2
,isobserved.
How-everfurtherinreaseupto500mW/m 2
dereasesthe
amplitudeofthisband.Thisresultisinterpretedasdue
to an inreasing arbon onentration in the samples.
ForlowRFpowerthesamplesaresilionrihwhilefor
high RFpowerarbonrih samplesareobtained. The
maximumintheSi-Cabsorptionbandobtainedfor250
mW/m 2
indiatesthatforthesedepositionparameters
veryloseto stoihiometrymaterial isprodued. The
observedinreaseinthearbonontentofthesamples,
also supported by the RBS measurements and by the
inreasing CH
n
related bands, indiates that for high
RFpowertheethingmehanismislesseÆientin
ex-trating theC-H bonds. This last result is onsistent
withtheobservedinreasein thedepositionrate.
Figure4. InfraredspetraforsamplesdepositedwithaH2
owof100sm,90%methanevolumefrationandvarying
theRFpowerdensity.
FromtheEXAFSdataattheSiKedgeitwas
pos-sible to analyze the short range strutural and
hem-ial order of thelms. Assumingthat thematerial is
a binary alloy, omposed of Si and C atoms, whih is
valid due to the low oxygen ontamination ( 1%
-3%),theoordinationofarbonatomswithsilionwas
estimated[15℄.
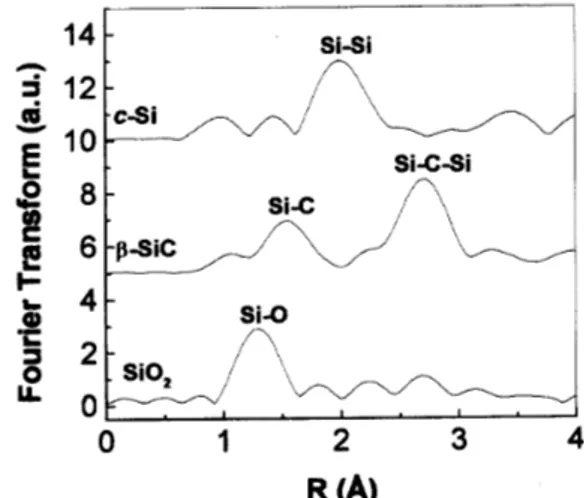
Figure5. Fouriertransformof theEXAFSspetrafor the
referenesamples.
Fig. 5 presents the Fourier transform of the
-SiC), showing the harateristi peaks. Due to the
phase-shift, theapparentbondingdistanes are about
1.15
AforSi-O,1.6
AforSi-C and2.0
AforSi-Si.
Figure6. Fouriertransformfor (a)thepowervariation
se-riesand(b)theH
2
dilutionvariationseries.
Fig. 6ashowstheFouriertransformoftheEXAFS
spetraof thesamplesgrownwithdierentRFpower.
ItisobservedthatsamplesgrownwithhigherRFpower
exhibit alargerareaunder thepeak assoiatedto the
Si-C oordination. Fig. 6b shows the Fourier
trans-form of the EXAFS spetra of samples deposited
un-der dierent H
2
dilution, at low RF power density
(12.5mW/m 2
). The data reveal that thesample
de-posited atthehighestH
2
dilution(300 sm) presents
amediumrangeorderstruture,withdistanesgreater
than3
A, relatedtotheseondoordinationshell.
TheoordinationfortheSi-Cbondinreasedfor
in-reasing RFpowerfrom 1.65,for sample deposited at
12.5 mW/m 2
, to 2.8 forthe sampledeposited at 125
mW/m 2
. ForallsamplesthetotalSioordinationwas
III.2. Doping
Theeletrialmeasurementsonn-typedoped
sam-plesonrm that the\starvingplasma" onditionhas
averyimportant eet onthe doping properties of
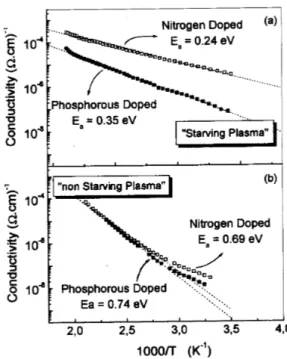
a-SiC:H. Fig. 7 shows the Arrhenius plots for samples
grown in \starving" and \non-starving" plasma
on-dition and implanted with 3.10 20
m 3
nitrogen and
phosphorus.
Figure 7. Arrhenius plots for nitrogen and phosphorous
dopedsamplegrownin(a)\starving"plasmaonditionand
(b)\non-starving"plasmaondition. Inbothasesthe
im-purityonentrationsis10 20
m 3
.
Itisobservedthatthesampleobtainedin\starving
plasma"showsanativation energy of0.24eV, whih
isnotonly alowvaluebut 3timeslowerthanthe
ex-hibitedbythesamplegrownin\non-starvingplasma"
onditions.Notethatthisbehaviorisobservedforboth
dopingelements,phosphorusandnitrogen,andthat
ni-trogenappearasamuhmoreeÆientdopingimpurity
thanphosphorus.
The results for samples grown in the silane
starv-ingplasmaonditionandimplantedwith10 20
and10 21
m 3
nitrogenandphosphorousaredepitedinFig. 8.
Foromparisontheeletrialondutivityforintrinsi
a-SiC:His inluded. As weansee, theresultsfor an
impurity onentrationof 10 21
m 3
are very
enour-aging, presenting an ativation energy as low as 0.12
eVandroomtemperaturedarkondutivityinthe
the \starving plasma" ondition improves the n-type
dopingeÆienyofthelmsuptolevelslosetothose
obtainedin deviequalitya-Si:H.
Figure8. N-typedoping: Arrheniusplotforstoihiometri
samples grown in \starving plasma" ondition implanted
with10 20
and10 2l
m 3
nitrogen(solidsymbol)and
phos-phorus(opensymbol).
Table2: Roomtemperatureeletrialondutivity(RT),
ondutivitytherrnalativationenergy(Ea)andthe
optial(Eo)fortheAldiusedsamples.
Also, it is observed that sample doped with 10 21
m 3
phosphorus has almost the sameativation
en-ergythansample dopedwith 10 20
m 3
nitrogen, but
exhibits a ondutivity signiantly lower, indiating
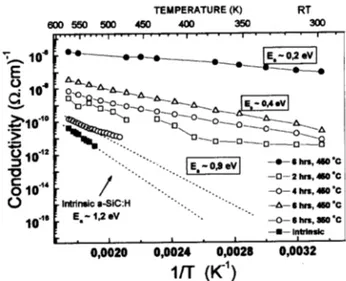
AsitanbeappreiatedinTable2,thealuminum
dif-fusionhadaleareetinthetransportharateristis
ofthesamples. Itisalsoobservedthat theoptialgap
remained unhanged for all samples. The Arrhenius
plot forthe aluminum diused samplesis depited on
Fig. 9.
As we an see, the doping was eetive, leading
to an ativation energy of 0,2 eV in the best ase,
whih is very low if we onsider that the energy gap
of thesamples is 2,4eV and that we aredealingwith
an amorphous alloy, where it is normally diÆult to
shift theFermi level position. Despitethe low
ativa-tion energy, the eletrial ondutivity remains quite
low,being10 7
(.m) 1
at roomtemperatureand
10 6
(.m) 1
at570K.Probablythisfatisrelated
with a non-ohmi harater of the metal ontats. It
isknownthatobtaininggoodohmiontatstop-type
SiC is a very diÆult task, even for rystalline
mate-rial,andspeiallyforlowerdopingonentrations. This
problem makesdiÆultto obtainreproduible results,
speially fortheabsolute valueof thedark
ondutiv-ity, sine the ontribution of the ontat resistaneis
notwellevaluated. It isimportantto reallherethat,
in general, the task of high ondutive p-type doping
andondentmetalontatstorystallineSiChasnot
yet beensolved [16℄. Inthis way, the resultsreported
herewithAldopingareverypromising.
Figure9. Condutivityvs. 1/Tfor intrinsiand Aldoped
samples. The data are tted with funtions ln = A i
+
B i
=T whereiis,Æ,,. Theativationenergiesarethe
slopesofthosefuntions.
IV Conlusions
Theresultsobtainedforsilionarbonalloyshave
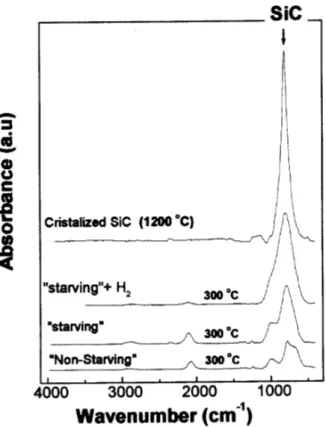
This fatiswellillustratedin Fig. 10whihshowsthe
FTIR spetrum for a sample rystallized at 1200 o
C
(rystallized SiC sample)together with the spetra of
three loseto stoihiometrya-SiC:Hasdeposited
sam-ples, grown in thefollowingonditions: (a) outof the
\silane starving regime" (\non-starving" sample), (b)
in\silanestarving"onditions(\starving"sample)and
() in \silane starving" onditions with hydrogen
di-luted gas mixture (\starving"+H
2
sample). It is
ob-served that the rystallized sample exhibits a single
peakspetrum,justonebandat800m 1
,
orrespond-ing toSi-C vibrations. The\starving"+H
2
sample
ex-hibits a large Si-C strething band and a very small
Si-H strething band. In fat, the spetrum is very
losetothatoftherystallizedsample,themain
dier-ene beingthefullwidthat halfmaximum,due tothe
struturaldisorder. Thisindiatesthattheshortrange
order ofourmaterialis verysimilartorystallineSiC,
in otherwords,wehaveattainedourgoalofproduing
theamorphousounterpartof-SiC.
Figure 10. FTIR spetra for 3as deposited samples(300
Æ
C)andonethermallyrystallizedat1200 Æ
C.
On the other hand, the \non-starving" sample
presentsjust asmallSiCpeakandexhibitsquitelarge
bandsorrespondingtoSi-H
n
(640and2090m 1
)and
Si-CH
3
vibrations(1250 m 1
). On the ontrary, the
\starving" sample already presents the main features
of silion arbide materials, this is to say a large
Si-toSi-H
n
bondsand someSi-CH
3
vibrations,the CH
n
vibrationsinthe2900m 1
spetralregionarealso
ap-parent.
These FTIR results are orroborated by the
EX-AFS experiments whih lead to an inreased
oordi-nation number for Si-C bonds for higher RF power
densities, signature of a higher degree of short range
order. In this way it was demonstrated that
onven-tional PECVD tehnique an be used suessfully to
produeamorphoussilionarbonalloyswithexellent
struturalpropertiesprovidedthat aset of deposition
parameters,whih enabletoontroltheplasma
hem-istry,areestablished.
It wasalso shown that the \starvingplasma"
on-dition has indeed an important role in inreasing the
n-typedoping eÆieny ofPECVD produed a-SiC:H
sineasampleimplantedwithanitrogen dose of10 20
m 3
grown in \non-starving" onditions showed an
ativation energy muh higher than a\starving"
pro-duedsampleimplantedwiththesamedose. F
urther-more,fora samplegrownin this ondition,ativation
energyanddarkondutivityvaluesomparableto
de-vie quality a-Si:H were obtained. It is also
demon-stratedthatnitrogendopingismoreeÆientthan
phos-phorus dopingfor these a-Si
1 x C
x
alloys. The
exper-iments with low temperature thermal diusion of Al
resulted in a very promising shift of the Fermi level.
Theeletrialondutivityhowever,mustbeinreased
and forthis purpose it is important to prepare ohmi
ontatsanddereasetheontatresistivity.
Aknowledgements
TheauthorsaregratefultoDr. M.H.Tabaniksfor
theRBSand FRSmeasurements. This work was
par-tially supported by the Brazilian agenies: FAPESP,
CNPqandCAPES.
Referenes
[1℄ J.Bullot and M.P. Shmidt, Phys.Stat. Sol. (b)143,
345(1987).
[2℄ R. S. Sussman and R. Odgen, Phil. Mag. B 44, 137
(1981).
[3℄ I. Solomon, M. P. Shmidt and H. Tran-Quo, Phys.
Rev.B38,9895 (1988).
[4℄ V.Mastelaro,A.M.Flank,M.C.A.Fantini,D.R.
Bitten-ourt,M.N.P.Carre~noandI.Pereyra,J.Appl.Phys.79,
1324(1996).
[5℄ I.Pereyra and M.N.P.Carre~no, Journal of Non-Cryst.
Solids201,110(1996).
[6℄ I.Pereyra,M.N.P.Carre~no, R.J.Prado,D.R.S.
[7℄ H.Wieder,M.CardonaandC.R.Guarnieri,Phys.
Sta-tusSolidiB92,99(1979).
[8℄ F. Evangelisti, J. of Non-Cryst. Sol. 164-166, 1009
(1993).
[9℄ C.C. Tsai,G.B.Anderson andR.Thompson,Mat.Res.
So.Symp.Pro.192,475(1990).
[10℄ Y.Hamakawa,D.Kruangam,T.Toyama,H.Yoshimi,
S. Paashe and H. Okamoto, Optoeletronis 4, 281
(1989).
[11℄ G.Ganguly,S.C.DE,SwatiRayandA.K.Barua,J.
ofNon-Crys.Sol.114,822(1989).
[12℄ M.N.P.Carre~no,J.P.Bottehia,I.Pereyra,ThinSolid
Films308-309,219(1997).
[13℄ M.N.P. Carre~no, I. Pereyra and H.E.M. Peres, J. of
Non-Cryst.Sol.227-230,483(1998).
[14℄ R.J. Prado, D.R.S. Bittenourt, M.H. Tabakniks,
M.C.A.Fantini,M.N.P.Carre~noandI.Pereyra,J.Appl.
Cryst.30,659(1997).
[15℄ G.S.CargillIIIandF.Spaepen,J.ofNon-Cryst.Solids
43,91(1981).
[16℄ J.Crofton,L.M.PorterandJ.R.Williams,Phys.Stat.