rev bras hematol hemoter. 2015;37(2):136–139
w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Case
Report
Remission
of
refractory
Crohn’s
disease
after
autologous
hematopoietic
stem
cell
transplantation
Milton
Artur
Ruiz
∗,
Roberto
Luiz
Kaiser
Junior,
Mikaell
Alexandre
Gouvêa
Faria,
Luiz
Gustavo
de
Quadros
Associac¸ãoPortuguesadeBeneficência,SãoJosédoRioPreto,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory: Received28July2014 Accepted27October2014 Availableonline30January2015
Introduction
Crohn’sdisease(CD)isaregionalenteritisthataffectsthe ter-minalileum,buthasthepotentialofinvolvinganysegment ofthegastrointestinaltractofthepatient.1
Theetiology ofCD isunknown, but severalproven risk factors exist such as family history, smoking, oral contra-ceptives,dietandethnicity.Thecombinationofriskfactors, andaberrantautoimmuneresponseintheintestinalmucosa withendothelialdysfunctionleadtodigestivetract malfunc-tioning.Diseasemanifestationsareheterogeneousbutmost patientspresent withabdominal pain, diarrheasometimes withblood,weightloss,feverandperianallesions.Upto30% ofCDpatientshavefistulae.
The current treatment is not ideal. Therapy generally involvessteroidsandanti-tumornecrosisfactoralpha(TNF␣)
blockersbutaboutone-thirdofpatientsfailtorespond (pri-marynon-responders)2and 10%ofpatientsdonottolerate
orareprimarynon-responders toalldrugsused.One-third of responders to anti-TNF␣ treatment show transient loss
∗ Correspondingauthorat:RuaCatarinaNucciParise,760,15090-470SãoJosédoRioPreto,SP,Brazil.
E-mailaddress:milruiz@yahoo.com.br(M.A.Ruiz).
ofresponse(secondarynon-responders).3 Severecasesmay
requirepartialresectionofasegmentofbowel,possiblytotal colectomywiththeplacementofstomabagsforfeceswhen the disease affects the rectum.1,4 Despite newtherapeutic
strategies, the number of surgeries remains stable.5 Many
patientsevolvewellwiththisapproach;however,sometimes the disease persists, becoming refractory and progressive, dramatically affecting the quality of life (QoL) of patients includingriskofdeath.6
Thus, new treatment options are needed. Recently, cell therapieswithTlymphocytes,tolerogenicdendriticcellsand bothhematopoieticandmesenchymalstemcells,havestarted toberesearched.7
Case
report
Thecaseofa28-year-oldfemalepatient isreportedwitha four-year diagnosis ofCD. She had been submitted to the resectionofa40-cmsegmentofbowelduetostenosis.Shewas refractory to treatment using corticosteroids, azathioprine,
http://dx.doi.org/10.1016/j.bjhh.2015.01.002
revbrashematolhemoter.2015;37(2):136–139
137
infliximaband adalimumab. Thepatient suffered repeated episodesofabdominalpain,mucusbloodydiarrhea,weight loss,depressionandpsychosis.
Shehadconstant,butmigratory,jointpainsince diagno-sis.Moreover,swellingofwristsandskinlesionsofalllimbs characteristicofpsoriaticarthritiswereapparentatthefirst examination.
An anatomopathological study of the antrum and cor-pusgastricumbyendoscopyandcolonoscopyshowedslight reactiveandvascularcongestionandaninvestigationof Heli-cobacterpyloriwasnegative.
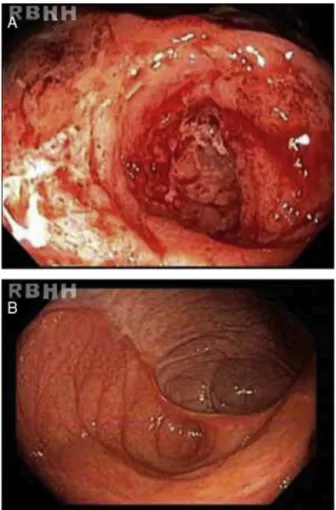
Active ileitis and proctitiswere observed in the ileoce-calvalve,rightcolon,left colonandrectum withextensive areasofulcerationandhistologicevidenceofinflammatory boweldiseasecompatible withCD.Additionally, changesin theregenerativearchitecturewereobservedaswere epithe-lioidgranulomaswithmultinucleatedgiantcellsbutwithout caseousnecrosis(Figure1).
Autologous hematopoietic stem cell transplantation (HSCT) was indicated. The procedure was only performed aftera court order asthis procedure is not authorized by the Brazilian healthcare system and after approval by the HospitalMedicalEthicsCommittee.
PriortostartingtheHSCTregimen,thepatientwas sub-mittedtophysicalandstandardlaboratoryevaluations.
TheCrohn’sDiseaseActivityIndex(CDAI)wasmorethan 450(severely ill)with largeulcerationsand involvementof morethan35%ofthesurface.8HerMontrealclassificationwas
A2/L3/B2with majorextraintestinal manifestations includ-ingiritistwoyearspreviouslyandpolyarticulararthritis.The patient’sQoLwasdeterminedusingtheQoLquestionnaire– ShortForm36(QoLSF-36).
The patient was mobilized with cyclophosphamide (60mg/kg) and granulocyte colony stimulating factor (10mg/kg) for seven days with 4.59×106 CD34+ cells/kg beingcollected from peripheralbloodin twoleukapheresis sessions (Hemonetics MSC+ USA). Thehematopoietic cells werecryopreservedindimethylsulfoxideandstoredinthe vaporphaseofliquidnitrogenuntiltransplantation.
Thepatientwasthenadmittedinthebonemarrow trans-plantunitwheresheunderwent aprophylaxisregimenfor parasitic,bacterial,andfungalagentsandPneumocystiscarinii. Ciprofloxacinwasadministeredforpartialbowel decontami-nationandthepatientunderwenthyperhydrationandmesna prophylaxistopreventhemorrhagiccystitis.The condition-ingregimenconsistedofcyclophosphamide(50mg/day)and rabbit anti-thymocyte globulin(Thymoglobulin, Genzyme – 1.25mg/kg/day)forfourdays.
Theneutrophilsdroppedbelow0.5×109cells/Lafteronly fivedaysandengraftmentoccurredonthe10thdayafter infu-sion.ThepatientwasdischargedonDay+12andtheonly tox-icityreportedwasmildmucositis.Transfusionrequirements wereonlytwobagsofirradiatedconcentratedredbloodcells. All the symptoms of the disease such as diarrhea and abdominal and joint pain disappeared in the mobilization periodbeforeASCT.
Cytomegalovirus(CMV)reactivationoccurredonDay+20, andwastreatedwithganciclovir(5mg/kg/day)for12days.
Three months after the HCST, the patient suffered a herpes zoster infection of the back; this improved
Figure1–(A)Serpiginousulcerations,withfold convergence,interveningridgesofswollencongested mucosaandcentralfibrinousexudate.(B)Whitescaring andscarretractionsevenmonthsafterautologous hematopoieticstemcelltransplantation.
within 15 days using specific local and systemic medica-tions.
TheresultsofasecondcolonoscopysixmonthsafterHSCT were normal (Figure 2). One year after the procedure the patientiswell,withoutanysymptomswithaCDAI<150 (clin-icalremission)andconsequentimprovementsinQoL.
Discussion
138
revbrashematolhemoter.2015;37(2):136–139Figure2–(A)Ileocolicanastomosiswithintense hyperemia,linearulcers,deformitiesandspontaneous friability.(B)Thesamesiteafterautologoushematopoietic stemcelltransplantationshowingcompletemucosal healing.
complicationrates havedroppeddue toexperience inthis procedure.Similartoseveralotherautoimmunediseases,risk shouldoutweighthepossibilityofcure.9Thus,HSCTisa
feasi-bleprocedure,which,whenincorporatedtotreatautoimmune diseases,canprovidegoodresultswithclinicalremission.The indicationofHSCTisbasedonimmunesystemablation fol-lowedbyregulationandregenerationoftheT-cellrepertoire viaimmunesystemresetting.10,11
Reports of HSCT for CD are not as common as some other diseases including multiple sclerosis and systemic sclerosis. Both autologous and allogeneic modalities have been described with consistent clinical and long term remission.12–14
Theindicationsoftheprocedurearenotwellestablished. However,HSCTshouldbeconsidered inspecificsevereand refractorycaseswhensurgicalresectionisindicatedandin patientswithahistoryofresection.Inthecurrentcase,the criteriaofthe EuropeanGroupforBoneMarrowTransplant (EBMT)werefollowedasdescribedbelow.15,16
AutologousHSCTshouldbeindicated:
• In severe cases that do not respond to treatment with immunosuppressiveagentsand anti-tumor necrosis fac-tors;
• InactiveorrefractoryCDthatisnotcontrolledby immuno-suppressantsorbiologicalagents;
• Whenthereisanindicationofsurgerybut withariskof smallbowelsyndrome;
• In refractorycolonic disease with perianal lesions when coloproctectomywithdefinitiveimplantationofthestoma isnotacceptedbythepatient.
In this case the hematologicand gastrointestinal toxic-itywasminimal.Symptomssuchasabdominalpain,profuse bloodydiarrhea,jointpainandcutaneouslesionsdisappeared eitherduringtheconditioning regimenor soonafterHSCT. Theclinical remissiondescribed inthis casehasremained untilthepresenttime,oneyearaftertheprocedure.
GlobalblockadeofimmunecellssuchasT-cellshasfailed inCDbuttherapiesusingmesenchymal,hematopoieticand adipose tissuesarepromising.17 Thebestideaseemstobe
autologousnon-myeloablativeHSCT.Inthelargestseriesso far, all 24 patients went into remission. Although a large numberofpatientsrelapseandrequiretreatmentafter trans-plantation, accordingtoBaumgart etal.the percentagesof patients in remission, steroid-free or medication-free five years after transplant were at least 70%, 80% and 60%, respectively.17
HSCTforCDinBrazilisnotaprocedureauthorizedbythe NationalHealthSystemorhealthinsuranceagencies.HSCT inthesesituationsisrestrictedtocasesenrolledinclinical trials,whichdonotexistinBrazilasthereisnoregulationfor compassionatetreatment.Thisprocedurewaspossibledueto acourtorderobtainedbythepatient,becauseoftheseverityof thecaseandcompliancetoEBMTcriteria.Therearenoother reportsoftheuseofHSCTforCDinBrazilbutthisprocedure seemstobesafeandwelltoleratedbythepatient.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.AbrahamC,ChoJH.Inflammatoryboweldisease.NEnglJ Med.2009;361(21):2066–78.
2.ColombelJF,SandbornWJ,RutgeertsP,EnnsR,HanauerSB, PanaccioneR,etal.Adalimumabformaintenanceofclinical responseandremissioninpatientswithCrohn’sdisease:the CHARMtrial.Gastroenterology.2007;132(1):52–65.
3.YanaiH,HanauerSB.Assessingresponseandlossof responsetobiologicaltherapiesinIBD.AmJGastroenterol. 2011;106(4):685–98.
4.HovdeO,MoumBA.Epidemiologyandclinicalcourseof Crohn’sdisease:resultsfromobservationalstudies.WorldJ Gastroenterol.2012;18(15):1723–31.
5.BernsteinCN,LoftusEVJr,NgSC,LakatosPL,MoumB. HospitalisationsandsurgeryinCrohn’sdisease.Gut. 2012;61(4):622–9.
revbrashematolhemoter.2015;37(2):136–139
139
7. Martínez-MontielMdelP,Gómez-GómezGJ,FloresAI. Therapywithstemcellsininflammatoryboweldisease. WorldJGastroenterol.2014;20(5):1211–27.
8. VilelaEG,TorresHO,MartinsFP,FerrariMA,AndradeMM, CunhaAS.EvaluationofinflammatoryactivityinCrohn’s diseaseandulcerativecolitis.WorldJGastroenterol. 2012;18(9):872–81.
9. SullivanKM,MuraroP,TyndallA.Hematopoieticcell transplantationforautoimmunedisease:updatesfrom EuropeantheUnitedStates.BiolBloodMarrowTransplant. 2010;16(Suppl1):S48–56.
10.MuraroPA,DouekDC,PackerA,ChungK,GuenagaFJ, Cassiani-IngoniR,etal.Thymicoutputgeneratesnewand diverseTCRrepertoireafterautologousstemcell
transplantationinmultiplesclerosispatients.JExpMed. 2005;201(5):805–16.
11.AlexanderT,ThielA,RosenO,MassenkeilG,SattlerA,Kohler S,etal.Depletionofautoreactiveimmunologicmemory followedbyautologoushematopoieticstemcell transplantationinpatientswithrefractorySLEinduces long-termremissionthroughdenovogenerationofajuvenile andtolerantimmunesystem.Blood.2009;113(1):214–23.
12.BurtRK,CraigRM,MilanettiF,QuigleyK,GozdziakP,BuchaJ, etal.Autologousnonmyeloablativehematopoieticstemcell transplantationinpatientswithsevereanti-TNFrefractory Crohndisease:long-termfollow-up.Blood.
2010;116(26):6123–32.
13.HommesDW,DuijvesteinM,ZelinkovaZ,StokkersPC,Ley MH,StokerJ,etal.Long-termfollow-upofautologous hematopoieticstemcelltransplantationforsevererefractory Crohn’sdisease.JCrohnsColitis.2011;5(6):543–9.
14.HasselblattP,DrognitzK,PothoffK,BertzH,KruisW,Schmiidt C,etal.RemissionofrefractoryCrohn’sdiseasebyhigh-dose cyclophosphamideandautologousperipheralbloodstemcell transplantation.AlimentPharmacolTher.2012;36(8):725–35.
15.SnowdenJA,SaccardiRA,AllezRM,ArdizzoneS,ArnoldR, CerveraR,etal.Hematopoieticstemcelltransplantationin severeautoimmunediseases:updatedguidelinesofthe EuropeanGroupforBloodandBoneMarrowTransplantation. BoneMarrowTransplant.2012;47(6):770–90.
16.HawkeyCJ.Stemcellsastreatmentininflammatorybowel disease.DigDis.2012;30(Suppl3):134–9.