w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Original
article
The
F309S
mutation
increases
factor
VIII
secretion
in
human
cell
line
Daianne
Maciely
Carvalho
Fantacini
a,b,∗,
Aparecida
Maria
Fontes
a,
Mário
Soares
de
Abreu
Neto
b,
Dimas
Tadeu
Covas
a,b,
Virgínia
Picanc¸o-Castro
a,baUniversidadedeSãoPaulo(USP),RibeirãoPreto,SP,Brazil bHemocentrodeRibeirãoPreto,RibeirãoPreto,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received29March2016 Accepted1April2016 Availableonline20April2016
Keywords:
HEK293cells
RecombinantfactorVIII F309Smutation Betaine
Sodium-4-phenylbutyrate
a
b
s
t
r
a
c
t
Objectives: ThecapacityofahumancelllinetosecreterecombinantfactorVIIIwithaF309S pointmutationwasinvestigated,aswastheeffectoftheadditionofchemicalchaperones (betaineandsodium-4-phenylbutyrate)onthesecretionoffactorVIII.
Methods:ThisworkusedavectorwithaF309SmutationintheA1domaintoinvestigateFVIII productionintheHEK293humancellline.FactorVIIIactivitywasmeasuredbychromogenic assay.Furthermore,theeffectsofchemicaldrugsontheculturewereevaluated.
Results:TheadditionoftheF309SmutationtoapreviouslydescribedFVIIIvariantincreased FVIIIsecretionby4.5fold.Moreover,theadditionofbetaineorsodium-4-phenylbutyrate increasedthesecretionrateofFVIIIBproteinsinHEK293cells,butthesameeffectwas notseenforFVIIIB-F309Sindicatingthatalltherecombinantproteinproducedhadbeen efficientlysecreted.
Conclusion: BioengineeringfactorVIIIexpressed inhumancellsmayleadtoanefficient productionofrecombinantfactorVIIIandcontributetowardlow-costcoagulationfactor replacementtherapyforhemophiliaA.FVIII-F309Sproducedinhumancellscanbeeffective
invivo.
©2016Associac¸ ˜aoBrasileiradeHematologia,HemoterapiaeTerapiaCelular.Published byElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-NDlicense
(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Hemophilia A is an inherited disorder linked to the X chromosomewhich resultsin deficiencyor abnormality of bloodcoagulationfactorVIII(FVIII).Thecurrenttherapyfor hemophiliaApatientsistheintravenousinfusionof plasma-derivedorrecombinantfactorVIII(rFVIII).Themajorproblem ofreplacement therapy with rFVIII is the low productivity
∗ Correspondingauthor.
E-mailaddress:[email protected](D.M.C.Fantacini).
which increases the cost of therapy. The high costs are mostlyattributabletoseveralbiochemicalcharacteristicsof therecombinantproteinsuchastheretentionofFVIIIwithin theendoplasmicreticulum(ER)duetoitsinteractionwith dif-ferentERchaperones.1
Severalstudieshaveprovidedimportantinformationthat helpedinthedesignanddevelopmentoftechniquesto bio-engineerrFVIII withbettersecretionefficiency.2,3 Swaroop4
developedanrFVIIIwithapointmutationintheA1domain
http://dx.doi.org/10.1016/j.bjhh.2016.04.002
(Phe309Ser) to increase secretion ofthe protein.4 However
despitemanysignificantadvancesintheheterologous expres-sionofFVIII,alowproductionratepersists.
Recently, it was suggested that recombinant concen-trates might be associated with a higher incidence of inhibitor development.5 This may be associated with the
different glycosylation pattern found in hamster cell lines [available productsareproduced inChinese hamsterovary (CHO) and Baby hamster kidney (BHK) cells] compared to humancells:Gal·1-3Gal1-(3)4GlcNAc(␣-Gal)epitopesand N-glycolylneuraminicacid(Neu5Gc)arenotpresentinhumans.6
Asanalternative,humancell lines canbeused to pro-duce recombinant coagulation factors. This heterologous expression system is capable of producing proteins with post-translationalmodificationssimilartotheiroriginal coun-terpartwhichmightreducethepossibilityofimmunogenic reactions.6
ThisstudyinvestigatedtheproductionofFVIIIcontaining theF309SpointmutationintheHEK293humancelllineand theefficacyofthemoleculeinvivoinahemophiliaAmouse model.
Methods
Lentiviralvectors
Ahumanimmunodeficiency virus-1(HIV-1)-basedlentiviral vector containing the human FVIII cDNA with a deletion of a large portion of the B domain (FVIIIB) and a sec-ondvectorwiththe Phe309Sermutation(A1 domain)were usedtoincreasethesecretionoftheprotein.Thetransgene expressionofbothvectorswasdrivenbytheinternal myelo-proliferative sarcoma virus (MSV) promoter with selection usingtheneomycingene.Thevectorswerekindlyprovided byDanielGibsonfromtheCraigVenterInstitute.
Transientexpression
FVIIIBandFVIIIB-F309Sconstructsweretransfected(10g ofDNA)intoHEK293cellsusinglipofectamine®2000reagent
(LifeTechnologies)followingthemanufacturer’sinstructions. Conditioned mediumwas harvested at48h and 72h after transfectionandanalyzedusingAsserachrom®VIII:Ag
(Diag-nosticaStago).
Productionoflentiviralparticles
Togeneratelentiviralparticles,theconstructDNAswere tran-sientlyintroduced into293FTcells bytripleco-transfection with the packaging construct pCMVR8.91 encoding gag, pol, and rev and the pseudotyping construct pMD2.VSVG codingforthe vesicularstomatitis virusglycoprotein (VSV-G). Transfection of plasmid DNAs was performed using lipofectamine® (Life Technologies) following the
manufac-turer’sinstructions.Viralparticleswereharvestedat48hand 72h post-transfection and filtered through a 0.22m filter (Millex®-GV).Theviralparticleswereconcentratedby
ultra-centrifugation(1.40h at31,000×g). Theconcentrated virus wasstoredat−80◦C.
Lentiviral titers were determined by quantitative poly-merasechainreaction(PCR)ofgenomicDNAfromtransduced 293FTcells.Briefly,1×105293FTcellswereseededinasix-well
plate andtransducedwithserialdilutionsofvector supple-mented with 8g/mL polybrene (Sigma-Aldrich). Genomic DNAwasextractedfromtransducedcells72hafter transduc-tionusingtheDNeasyBloodandTissueKit(Qiagen)asperthe manufacturer’s instructions, and quantitativePCRwas car-riedoutinduplicateonsamplestodeterminebothtotalviral DNAandhuman-actinlevels(ACTB).Theintegratingcopy number per transducedcell wasnormalized assumingtwo ACTBallelespercellandinfectiousunitspermilliliterwere calculated as total viral DNA/(ACTB/2)×(1×105)×dilution
factor. Primers and probe used have been described previously.7
ExpressionofstableHEK293cells
HEK293cellsweremaintainedinDulbecco’smodifiedeagle medium(DMEM–Life Technology)supplementedwith10% (v/v) fetal bovine serum (ThermoScientific), 1%(v/v) peni-cillin/streptomycinsolution(Gibco)in5%CO2at37◦C.Cells
wereplatedonto24-wellplates(approximately100,000cells) andtransducedatamultiplicityofinfection(MOI)of40for transduction 1–4,MOIof80fortransduction5, andMOIof 160fortransduction6–10withviralparticlesinafinalvolume of500LofDMEMsupplementedwith8g/mLofpolybrene (Sigma). At 24h post-transduction, virus-containing media was replaced with fresh DMEM and cells were allowed to recover by culturing overnight at 37◦C in 5% CO
2.
Subse-quently, transduced cells were replaced foranother round oftransductionuntilthetenthcycleand analyzedforFVIII activity,copynumberintegration,transcriptexpressionand proteinanalysis.
FactorVIIIquantificationandantigenanalysis
FVIIIactivitywasdeterminedbyatwo-stageassayusingthe COAMATIC FVIII (Chromogenix) according tothe manufac-turer’s instructions. Normal human reference plasma was usedtogeneratethestandardcurve.TheFVIIIantigenwas quantifiedbyanenzyme-linkedimmunosorbentassay(ELISA) methodusingtheAsserachromVIII:Ag(Stago)commercialKit accordingtothemanufacturer’sinstructions.
Reversetranscriptionpolymerasechainreactionanalysis
RNAwasextractedusingtheRNeasyMiniKit(Qiagen) accord-ing tothemanufacturer’sinstructions.Quantitative reverse transcription-polymerase chain reaction (RT-PCR) was per-formedusingtheHighCapacitycDNAreversetranscriptionKit (LifeTechnology).ThiswasfollowedbyPCRwithprimersBiPfor
(5′-CCAACGCCAAGCAACCAAAG-3′),BiP
rev(5′-CTTCTCCCC
CTCCCTCTTAT-3′),GAPDH
for(5′-GCCTCAAGATCATCAGCA
ATGC-3′),GAPDHrev(5′-CATGGACTGTGGTCATGAGTC
CT-3′).SYBRGreenreal-timePCRwascarriedoutusingABIPrism
theamountofappliedRNA.Relativeimmunoglobulin-binding protein(BiP)transcriptionlevelsweremeasuredbyapplying the2−(Ct)equation.
Westernblotanalysis
Conditionedmediumwascollectedfromtransducedand non-transduced HEK 293 cells after 72h and total protein was quantifiedbytheBCAProteinAssayKit(Pierce).Thecultured cellswere lysedinice-coldlysis buffercontainingprotease inhibitors(Completemini-proteaseinhibitorcocktail,Roche) and centrifuged at 16,000×g for 15min. Total proteinwas quantifiedbythe BCA ProteinAssay Kit(Pierce).Total pro-tein(35gand40gextractedfromconditionedmediumand lysedcells,respectively)wasseparatedbySDS-PAGE(4–20% Mini-PROTEAN,BIO-RAD),transferredtonitrocellulose mem-branes(40m, Hybond-CExtra,AmershamBioscience)and probed with anti-FVIII light chain mouse antibody (Santa Cruz Biotechnology)or anti-BiP/GRP78 mouse antibody(BD Bioscience).Internalcontrolwasachievedusinganti-actin mouseantibody(Sigma-Aldrich).
Chemicalchaperonetreatmentoftransducedcells
Transduced cells were seeded at a cell density of about 3.5×106cellsperplateandwereincubated inthepresence
ofchemicalchaperones:betaine(Sigma-Aldrich)andsodium 4-phenylbutyrate(Sigma-Aldrich)atdifferentconcentrations. After72hofincubationwithchemicalchaperones,levelsof active FVIII were determined in the supernatants. Experi-mentswereperformedinduplicate.
Invivoassay
HemophiliaAmice(B6;129S4-F8tm1kaz/J)were obtainedfrom JacksonLaboratory(BarHarbor,ME, USA).Adultmice,aged 8–12weeks, were used fortail clipping. All protocols were conductedinaccordance withthe EthicalCode forAnimal Experimentationofthe CouncilforInternational Organiza-tionsofMedicalSciences(CIOMS)andtheColégioBrasileirode Experimentac¸ãoAnimal(COBEA).Thisstudywasapprovedby theEthicsCommitteeforAnimalResearchoftheUniversidade deSãoPaulo(USP),RibeirãoPreto(#14.1.784.53.8).
Severetail-bleedingmodel
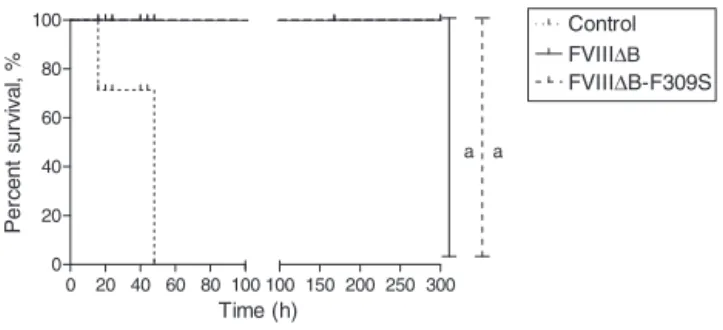
Thetail-clipchallenge wasdesignedtoanalyze hemophilic A mice after receiving one unit of culture supernatant containing FVIIIB-F309S (n=3 mice), FVIIIB (n=3 mice) or phosphate-buffered saline (PBS) (n=3 mice). Mice were anesthetizedwith4.5%isofluraneandreceivedculture super-natantviaretroorbitalinjection.After10min,bleedingwas inducedbycutting1cmofthetailandanimalsurvivalwas monitoredover50h.
Statisticalanalysis
Resultsareexpressedasmeans±standarderrorofthemean (SEM) or standard deviation (SD) as appropriate. Student’s unpaired t-testand 95% confidenceinterval were used for
comparisonsbetweenthegroups.Two-wayanalysisof vari-ance(ANOVA)wasusedtocomparedifferencesbetweenthe indicatedgroupsandthelog-ranktestwasusedtocompare thesurvivalcurvesafterthetail-clippingchallenge.p-Values <0.05wereconsideredstatisticallysignificant.
Results
Transientassay
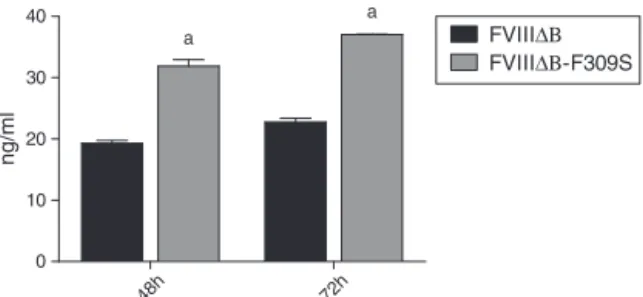
InordertostudytheroleoftheF309Smutationonthe secre-tionandfunctionofFVIIIinhumancelllines,amoleculewith thismutationwassynthesizedandclonedintoalentiviral vec-tor.After,itssecretionwasanalyzedbytransienttransfection inHEK293cells.FVIIItwo-stageactivityassaysperformedon conditionedmediaharvested48hand72haftertransfection revealed thattherewasabettersecretionofFVIIIB-F309S overtheFVIIIBlevels(Figure1).
TheFVIIIB-F309S presentedon average, 1.5and 2fold highersecretionthanFVIIIBinHEK293cells,after48hand 72hoftransfection.Theseresultswerereproducibleand con-sistentinthreeseparatetransfectionexperiments.
GenerationoftheHEK293celllinestablyexpressing FVIIIBandFVIIIB-F309S
To compare whether the mutation could also result in increasedsecretionofFVIIIinstableexpression,HEK293cell linesstablyexpressingFVIIIBandFVIIIB-F309Swere gen-erated.Thesecelllinesweregeneratedbylentiviralinfection followedbyselectionusinggeneticin.Thenumberof lentivi-ralvectorsintegratedintothegenomewerequantifiedforthe twogeneratedstrains(Figure2A).ThecelllineFVIIIB-F309S hasonecopy/cellofthelentiviralvectorwhereastheFVIIIB has1.4copies/cellintegratedinitsgenome.
Thecell linecontaining theFVIII mutationexpresses 9-foldmoreFVIIImRNAthanthecellwithFVIIIB(Figure2B). ThelevelofproteinsecretedinthecellwithFVIIIB-F309S isgreaterthanthecell linewithFVIIIB.Thecellline con-tainingtheF309SmutationsecretedanaverageoffiveIU/106
cells/72hofFVIII,thatis,4.5-foldmoreefficientlythanFVIIIB asdeterminedbytwo-stageclottingactivityassay(Figure2C).
48h 72h
0 10 20 30 40
FVIIIΔΒ FVIIIΔΒ-F309S
ng/ml
a
a
Figure1–TransientproductionofFVIIIBand
FVIIIB-F309SinHEK293cells.ThefactorFVIIIproduction wasmeasuredbytheenzyme-linkedimmunosorbent assay(ELISA)technique.aStatisticallysignificant
A
B
C
REU
UI/10
6 cells/72h
800 000.0
p-value=0.0216
p-value=0.0012
600 000.0
400 000.0
200 000.0
0.0
6
4
2
0
Cells Vector copy/cell
FVIIIΔB-F309S
FVIIIΔB-F309S FVIIIΔB FVIIIΔB-F309S FVIIIΔB 1
FVIIIΔB 1.4
Figure2–(A)Numberoflentiviralvectorsintegratedinto genome;(B)relativeexpressionunits(REU)ofboth recombinantFVIIIexpressionsand(C)FactorVIII
second-stageactivitybyachromogenicassay(COAMATIC). Datapresentedarethemeanofthreeindependent experimentsandtheerrorbarsrepresentthestandard deviation.Asterisksdesignatestaticallysignificant differencesbetweengroupsusingtwo-wayANOVA
CharacterizationofsecretedFVIIIB-F309S
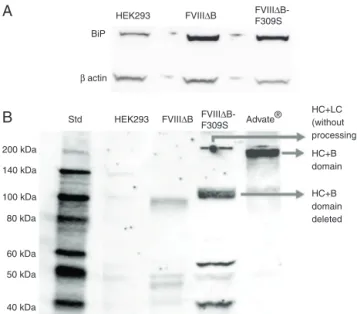
FVIII protein has a 110-amino acid region within the A1-domainthatinhibitsitssecretionandcontainsmultipleshort peptide sequences that have potential to bind to BiP. The low level of FVIII secretion correlates with binding to the BiP,withinthelumenoftheER.Toassesswhetherthehigh expression of FVIII induces the production of BiP, the BIP levelwasevaluatedbyWesternblot.Figure3showsthatFVIII
HEK293
BiP
Advate®
HC+LC (without processing)
HC+B domain deleted HC+B domain β actin
Std
200 kDa
A
B
140 kDa
100 kDa
80 kDa
60 kDa
50 kDa
40 kDa
HEK293
FVIIIΔB
FVIIIΔB
FVIIIΔ B-F309S
FVIIIΔ B-F309S
Figure3–CharacterizationofFVIIIproducingcelllinesby Westernblot.(A)Celllysateswerefractionatedby SDS–PAGE,andimmunoblottedwithBiP(78kDa)and b-actin(42kDa)antibodies.(B)Supernatantsof
non-transducedHEK293cells,293cellstransducedwith FVIIIBandFVIIIB-F309S,andthecontrolAdvate®
immunoblottedwithanti-heavychainFVIII.
expressionleadstoanincreasedamountofBiP.However,there arenosignificantdifferencesinBiPamountsbetweenHEK293 cellsexpressingFVIIIBandthoseexpressingFVIIIB-F309S (Figure3A).
InordertocharacterizethesecretedFVIIIr,Westernblot wasperformedwiththeanti-heavychainmonoclonal anti-body.Asexpected,theHEK293cellsexpressingFVIIIB-F309S secretemoreFVIIIthanHEK293cellsexpressingFVIIIB.It waspossibletodetecta90kDabandreferring totheheavy chain(Figure3B).
However,itwasnotpossibletoidentifyclearlytheband referring to the heavy chain in the FVIIIBsample, prob-ably because this cell produces a low amount of FVIII thereby hindering antibodybinding. Inthe band relatedto theheavychain,anotherbandof∼200kDawasdetectedin commercial FVIII corresponding to theheavy chainwithB domain(Advate®,Baxter).FVIIIB-F309S,whichhasthe
par-tial deletion ofB domain,alsoshows a band of∼200kDa, suggesting that part of the FVIIIr released is composed of heavy and light chains without intracellular cleavage (Figure3B).
The FVIIIB-F309S sample also showed fragments of 53kDaand43kDa,whichareprobablyrelatedtothe degra-dationofFVIII.The53kDabandmaycorrespondtopartofthe A2domainwiththeA1domain(∼55kDa)andthe43kDais relatedtotheA2domain(Figure3B).
Chemicalchaperonesupplementationincreasesthe secretionofFVIIIBbutnotofFVIIIB-F309S
Inaddition,twodifferentcompoundstermedchemical chap-erones (CC)were tested. Thesecompounds are responsible fornon-specificallystabilizingnative proteinconformations and supporting escape from the endoplasmic quality con-trol system. Members ofdifferentcompound classes ofCC (Betaineandsodium-4-phenylbutyrate)weretestedfortheir effecton FVIIIBand FVIIIB-F309SexpressioninHEK293 cells.Betaineandsodium-4-phenylbutyratewereusedat con-centrations of 1mM and 100mM. The drugs were tested individually and in combination in the modified cell lines expressingFVIIIBandFVIIIB-F309S.
The addition of betaine or sodium-4-phenylbutyrate increasedthesecretionrateofFVIII-BproteinsinHEK293 cells(Figure4).Thecombinationofbothdrugsledtoafurther increaseinthesecretionofFVIIIB.However,noincreasein thesecretionofFVIIIB-F309Swasobservedwiththeaddition ofbetaineorsodium-4-phenylbutyrateorwiththe combina-tionofthesetwocompounds(Figure4).
FVIIIBandFVIIIB-F309Sarefunctionallyactivein hemophiliaAmice.
4
a a a
b b
Control Betaine 100 mM
Sodium-4-phenylbutyrate 1 mM
Sodium-4-phenylbutyrate 1 mM Betaine 100 mM+
3
2
F
old change
1
0
FVIII ΔB
FVIII ΔB-F309S
Figure4–Effectofthebetaineand
sodium-4-phenylbutyrateonFVIIIsecretion.HEK293cells expressingFVIIIBorFVIIIB-F309Swereincubatedwith betaineandsodium-4-phenylbutyratealoneorin
combination.FVIIIactivitywasdeterminedincell supernatantsafter72hbychromogenicassay(n=3).
aStatisticallysignificantdifferencesbetweengroupsusing
two-wayANOVA.p-Value=0.0045.bStatisticallysignificant
differencesbetweengroupsusingtwo-wayANOVA. p-Value=0.0005.
0 20 40 60 80100 0
20 40 60 80 100
100 150200 250 300
Control FVIIIΔB FVIIIΔB-F309S
a a
Time (h)
P
e
rc
e
n
t s
u
rv
iv
a
l, %
Figure5–SurvivalcurveofhemophiliaAmicesubmitted totreatmentwithFVIII-B(n=4),FVIIIB-F309S(n=4)or phosphate-bufferedsaline(n=4)andchallengedbytail clipping.aStatisticallysignificantvalues(p-value=0.0136).
Discussion
RecombinantFVIIIproteinisoneofthemostcomplex pro-teinsforindustrialproductionduetothelowefficiencyofgene transcription,proteininteractions withretentionintheER, inappropriatetransportfromtheERtotheGolgiapparatusand theinstabilityofthesecretedprotein.8–10Overtheyears,other
researchgroupshavestudiedwaystoimprovetheexpression, secretionandtoincreasethehalf-lifeofcoagulationfactors, especiallyFVIII.2,11
Inthisstudy,theHEK293celllinewaschosenduetothe variousadvantagesofferedbythisstrain,forexample,robust patternofgrowth,easymaintenance,andhightransfection efficiency and productionof proteins.12 The transient
pro-ductionofrFVIII using theserum-free HEK293cell line in suspensionhasbeen demonstrated yielding approximately 0.64IU/mLFVIIIr.13Morerecently,thepharmaceutical
indus-try hasdeveloped the first rFVIII producedin humancells (usingtheHEK293Fstrain)whichshowedefficiencyinterms ofsafetyandproduction.14
ThecurrentstudyshowedthattheF309Smutationwasable toincreaseFVIIIsecretionby3-foldcomparedtoFVIIIBin
astableHEK293cellline.Thisresultisinaccordancewith previousworks.2,4Itisknownthattheinefficientsecretionof
FVIIIiscorrelatedwithbindingtotheproteinidentifiedasBiP, alsoknownastheglucose-regulatedprotein78(GRP78)within thelumenoftheER.15Marquetteetal.locateda110amino
acidregionwithintheA1domainthatinhibitsFVIIIsecretion. Thisregionisclusteredwithmultipleshortpeptidesequences thathavepotentialtobind toBiP.16 TheresultsofWestern
BlotinthisstudyrevealedthatbothFVIIIB-F309SandFVIIIB increasedBiPexpression,althoughFVIIIproductionwasnot affected.
AnotherstrategyusedinthisworktoimproveFVIII secre-tionwastoaddchemicalchaperonestoculturecells.Betaine isachemicalchaperonethatcaninhibittheaggregationof FVIIIandrestoretheintracellulartraffickingoflooselycoiled proteins.Inadditiontobetaine,sodiumbutyrate,anorganic compoundthathasvariouseffectsoncellcultures,wasalso tested;oneeffectistheinductionofgeneexpressionby his-tone hyperacetylation promotingthe activation ofgenes.17
This study shows that HEK 293 cells expressing FVIIIB that had been treated with betaine and sodium butyrate increasedFVIIIproduction.However,noeffectwasobservedin HEK293cellsexpressingFVIIIB-F309Streatedwithbetaine. Roth18testedseveralchemicalchaperones,includingbetaine
and reported that this compound increased the secretion of FVIII because ofthe increasedsolubility ofintracellular FVIIIaggregatesandimprovedtransportfromtheERtothe Golgi apparatus.18 By contrast, the data ofthis study
sug-gestthatalltheFVIIIproducedbyHEK293cellsexpressing FVIIIB-F309Swassecretedoutofthecellanddidnotform aggregates.
Ontheotherhand,sodiumbutyrateincreasedFVIII pro-ductioninHEK293cellsexpressingFVIIIBeitheralone or incombinationwithbetaine.Previousstudieshaveshowed that sodiumbutyrateproducesawidevarietyofeffectson cellsinculture:arrestofcellgrowth,reversionofthe trans-formationcharacteristicsofcells,andinductionofproteins, including enzymes, peptide hormones and hemoglobin.17
However,the sameeffectwas notnoticedinHEK293 cells expressing FVIIIB-F309S with the reason for this differ-ence not being clear and thus further investigations are needed.
Conclusion
ThemaincontributionofthisworkistheproductionofaFVIII moleculewithahighsecretionrate(withtheF309Smutation intheA1domain),inordertoincreasetheproductivityand decreasetheproductioncost.Furthermore,thisstudyutilized ahumancellline,HEK293,toproduceanrFVIIImoresimilar to the existing FVIII inhuman plasma, and less immuno-genic than the rFVIII commercially produced in hamster cells.
Conflicts
of
interest
Acknowledgements
WethankCleideAraújoSilvaandSandraNavarroBresciani whohelpedwiththeanimalexperimentsanddrewthefigures, respectively.ThisworkwassupportedbyCAPES,FAPESPand CNPq.
r
e
f
e
r
e
n
c
e
s
1. DornerAJ,WasleyLC,KaufmanRJ.Increasedsynthesisof
secretedproteinsinducesexpressionofglucose-regulated
proteinsinbutyrate-treatedChinesehamsterovarycells.J
BiolChem.1989;264(34):20602–7.
2. MiaoHZ,SirachainanN,PalmerL,KucabP,CunninghamMA,
KaufmanRJ,etal.BioengineeringofcoagulationfactorVIII
forimprovedsecretion.Blood.2004;103(9):3412–9.
3. DoorissKL,DenningG,GangadharanB,JavazonEH,McCarty
DA,SpencerHT,etal.ComparisonoffactorVIIItransgenes
bioengineeredforimprovedexpressioningenetherapyof
hemophiliaA.HumGeneTher.2009;20(5):465–78.
4. SwaroopM,MoussalliM,PipeSW,KaufmanRJ.Mutagenesis
ofapotentialimmunoglobulin-bindingprotein-bindingsite
enhancessecretionofcoagulationfactorVIII.JBiolChem.
1997;272(39):24121–4.
5. CalvezT,ChambostH,Claeyssens-DonadelS,d’OironR,
GouletV,GuilletB,etal.RecombinantfactorVIIIproducts
andinhibitordevelopmentinpreviouslyuntreatedboyswith
severehemophiliaA.Blood.2014;124(23):3398–408.
6. Picanco-CastroV,BiaggioRT,CovaDT,SwiechK.Production
ofrecombinanttherapeuticproteinsinhumancells:current
achievementsandfutureperspectives.ProteinPeptLett.
2013;20(12):1373–8.
7. MatsuiH,ShibataM,BrownB,LabelleA,HegadornC,
AndrewsC,etal.ExvivogenetherapyforhemophiliaAthat
enhancessafedeliveryandsustainedinvivofactorVIII
expressionfromlentivirallyengineeredendothelial
progenitors.StemCells.2007;25(10):2660–9.
8. SoukharevS,HammondD,AnanyevaNM,AndersonJA,
HauserCA,PipeS,etal.ExpressionoffactorVIIIin
recombinantandtransgenicsystems.BloodCellsMolDis.
2002;28(2):234–48.
9.ChuahMK,VandendriesscheT,MorganRA.Developmentand
analysisofretroviralvectorsexpressinghumanfactorVIIIas
apotentialgenetherapyforhemophiliaA.HumGeneTher.
1995;6(11):1363–77.
10.BeckerS,SimpsonJC,PepperkokR,HeinzS,HerderC,GrezM,
etal.Confocalmicroscopyanalysisofnative,fulllengthand
B-domaindeletedcoagulationfactorVIIItraffickingin
mammaliancells.ThrombHaemost.2004;92(1):23–35.
11.DumontJA,LiuT,LowSC,ZhangX,KamphausG,SakorafasP,
etal.ProlongedactivityofarecombinantfactorVIII-Fcfusion
proteininhemophiliaAmiceanddogs.Blood.
2012;119(13):3024–30.
12.ThomasP,SmartTG.HEK293cellline:avehicleforthe
expressionofrecombinantproteins.JPharmacolToxicol
Methods.2005;51(3):187–200.
13.SwiechK,KamenA,AnsorgeS,DurocherY,Picanc¸o-CastroV,
Russo-CarbolanteEM,etal.Transienttransfectionof
serum-freesuspensionHEK293cellcultureforefficient
productionofhumanrFVIII.BMCBiotechnol.2011;11:
114.
14.ValentinoLA,NegrierC,KohlaG,TiedeA,LiesnerR,HartD,
etal.ThefirstrecombinantFVIIIproducedinhumancells–
anupdateonitsclinicaldevelopmentprogramme.
Haemophilia.2014;20Suppl.1:1–9.
15.MunroS,PelhamHR.AnHsp70-likeproteinintheER:identity
withthe78kDaglucose-regulatedproteinand
immunoglobulinheavychainbindingprotein.Cell.
1986;46(2):291–300.
16.MarquetteKA,PittmanDD,KaufmanRJ.A110-aminoacid
regionwithintheA1-domainofcoagulationfactorVIII
inhibitssecretionfrommammaliancells.JBiolChem.
1995;270:10297–303.
17.KruhJ.Effectsofsodiumbutyrate,anewpharmacological
agent,oncellsinculture.MolCellBiochem.1982;42(2):
65–82.
18.RothSD,SchüttrumpfJ,MilanovP,AbrissD,UngererC,
Quade-LyssyP,etal.Chemicalchaperonesimproveprotein
secretionandrescuemutantfactorVIIIinmicewith