REVISTA
PAULISTA
DE
PEDIATRIA
www.rpped.com.br
CASE
REPORT
Hirschsprung’s
disease
---
Postsurgical
intestinal
dysmotility
Mariana
Tresoldi
das
Neves
Romaneli,
Antonio
Fernando
Ribeiro,
Joaquim
Murray
Bustorff-Silva,
Rita
Barbosa
de
Carvalho,
Elizete
Aparecida
Lomazi
∗FaculdadedeCiênciasMédicasdaUniversidadeEstadualdeCampinas(Unicamp),Campinas,SP,Brazil
Received13August2015;accepted22December2015 Availableonline12May2016
KEYWORDS
Infant; Hirschsprung’s disease; Gastrointestinal motility
Abstract
Objective: TodescribethecaseofaninfantwithHirschsprung’sdisease presentingastotal colonicaganglionosis,which,aftersurgicalresectionoftheaganglionicsegmentpersistedwith irreversiblefunctionalintestinalobstruction;discussthedifficultiesinmanagingthisformof congenitalaganglionosisanddiscussaplausiblepathogeneticmechanismforthiscase.
Casedescription: The diagnosis ofHirschsprung’s disease presenting as total colonic agan-glionosis was established in a two-month-old infant, after an episode of enterocolitis, hypovolemicshockandseveremalnutrition.Aftercolonicresection,thepatientdidnotrecover intestinalmotorfunctionthatwouldallowenteralfeeding.Postoperativeexaminationof rem-nantileumshowedthepresenceofganglionicplexusandareducednumberofinterstitialcells ofCajalintheproximalbowelsegments.At12months,thepatientremainsdependentontotal parenteralnutrition.
Comments: Hirschsprung’sdisease presentingastotal colonicaganglionosis hasclinicaland surgicalcharacteristicsthatdifferentiateitfromtheclassicforms,complicatingthediagnosis andtheclinicalandsurgicalmanagement.Thepostoperativecoursemaybeassociatedwith permanentmorbidityduetointestinaldysmotility.The numericalreductionoralterationof neuralconnectionsintheinterstitialcellsofCajalmayrepresentapossiblephysiopathological basisforthecondition.
©2016SociedadedePediatriadeS˜aoPaulo.PublishedbyElsevierEditoraLtda.Thisisanopen accessarticleundertheCCBYlicense(http://creativecommons.org/licenses/by/4.0/).
∗Correspondingauthor.
E-mails:[email protected],[email protected](E.A.Lomazi). http://dx.doi.org/10.1016/j.rppede.2016.05.001
PALAVRAS-CHAVE
Lactente; Doenc¸ade Hirschsprung; Motilidade gastrointestinal
Doenc¸adeHirschsprung---Dismotilidadeintestinalpós-cirúrgica
Resumo
Objetivo: Descreverocasodeumlactenteportadordedoenc¸adeHirschsprungnaforma agan-glionose colônica total que,apósaressecc¸ãocirúrgicado segmentoagangliônico,manteve quadroirreversíveldeobstruc¸ãointestinalfuncional;discutirasdificuldadesnomanejodessa formadaaganglionosecongênitaeapontarummecanismopatogenéticoplausívelparaocaso.
Descric¸ãodocaso: Odiagnósticodedoenc¸adeHirschsprungnaformaaganglionosecolônica totalfoidefinidoemlactenteaosdoismesesdevida,apósepisódiodeenterocolite,choque hipovolêmicoedesnutric¸ãograve.Apósressecc¸ãocolônica,opacientenãorecuperouafunc¸ão motora intestinal que possibilitasse a alimentac¸ão viaenteral. Oexame do íleo remanes-centepós-operatóriomostroupresenc¸adeplexosganglionaresereduc¸ãonuméricadascélulas intersticiaisdeCajalemsegmentosproximaisdointestino.Aos12mesesdevida,opaciente mantém-sedependentedenutric¸ãoparenteraltotal.
Comentários: Adoenc¸adeHirschsprungnaformaaganglionosecolônicatotaltem particular-idadesclínico-cirúrgicasqueadiferenciamdasformasclássicasedificultamodiagnósticoeo manejoclínico-cirúrgico.Aevoluc¸ãopós-operatóriapodeassociar-seàmorbidadepermanente decorrentededismotilidadeintestinal.Areduc¸ãonuméricaouasalterac¸õesdasconexões neu-raisdascélulasintersticiaisdeCajalpodemrepresentarumapossívelbasefisiopatológicapara acondic¸ão.
©2016SociedadedePediatriadeS˜aoPaulo. PublicadoporElsevier EditoraLtda.Este ´eum artigoOpenAccesssobumalicenc¸aCCBY(http://creativecommons.org/licenses/by/4.0/).
Introduction
Hirschsprung’sdisease(HD)isthemostprevalentcauseof functionalbowel obstructionin infants,withan incidence of1:5000livebirths.1Itisgeneticallydeterminedand
cha-racterizedbyadefectinthemigrationofembryoniccells
fromtheneuralcrest,generatinganaganglionicsegmentat
thedistalendoftheintestines.2Thegoldstandard
diagnos-ticmethodis arectalbiopsy showingabsenceofganglion
cellsandincreasednumberofacetylcholinesterase-positive
nervefibers.3
The anatomicallocationof the transition between the
distalaganglionicsegmentandtheproximalganglionic
seg-mentallowsfortheclassificationofHDasfollows:classic
---whentheaganglionicsegmentextendstotheproximal
sig-moid;withlongsegment---whenaganglionosisreachesthe
splenicflexureorthetransversecolon;ortotalcolonic
agan-glionosis(HDTCA)---whentheaganglionicsegmentextends
fromtheanusuptoat most50cmproximaltothe
ileoce-calvalve.HDTCApresentsclinical,histological,andgenetic
differencesinrelationtotheothertypesofHD,andis
asso-ciated withdiagnostic and management difficulties.4 The
classicformofHDisobservedin7---88.8%ofcases;thelong
form,in3.9---23.7%;andHDTCA,inupto12.6%ofpatients.5
Surgical therapy in HD minimizes the complications of
intestinalobstructionwhentheaganglionicsegmentis
com-pletelyresected.Insomepatients,postoperativeintestinal
dysmotilitypersists,mostoftenmanifestedaschronic
cons-tipation andrecurrent episodesof enterocolitis.Different
histopathologic findings can be identified in these cases,
such asincompleteresection of theaganglionic segment,
hypoganglionosis, and intestinal neuronal dysplasia
juxta-posedtotheaganglioniczone.4
Thepresentarticlereportsacaseinwhichdifficultiesof
diagnosis,therapy,andprognosiswereobserved.The
publi-cationofthiscasereportwasapprovedbytheInstitutional
ReviewBoard(IRB)oftheStateUniversityofCampinas,IRB
Opinion/ArticleNo◦012/2015fromJuly28,2015.
Case
description
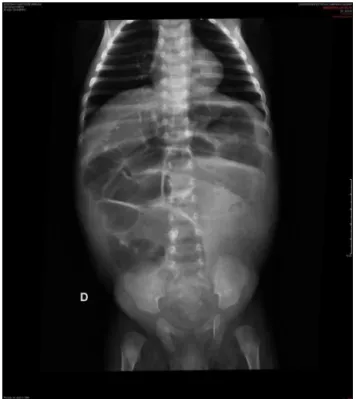
Ablackmalepatientwasreferredtoatertiaryhospitalat age2months,withadiagnosisofintractablediarrheaand vomitingfor22days.Heevolvedwithhypovolemicshockand refractorymetabolic acidosis. The patienthad undergone asimpleabdominalradiograph,whichshowedwidespread bowel distension(Fig. 1), and a CT scanshowing lack of
progressionoftheenteralcontrasttothedistalcolonic
seg-ments.
History of prenatal ultrasound showing distended fetal
bowelloops. The motherdenied delay (>24h) in the
pas-sageofmeconiumatbirthandcomplaintscompatiblewith
intestinalobstruction. The childwasborn vaginally,at 39
weeksofgestationalage,dischargedfromthehospitalon
thethirdday oflife,withbirthweightof 2950g.Neonatal
screenings for phenylketonuria, hypothyroidism, and
cys-ticfibrosis werenegative.Thechild hadbeen inexclusive
breastfeedingsincebirth;themotherdeniedconstipation,
but reportedpoor weight gain. After38 days of life, the
childhaddiarrheathatrequired hospitalizationinthecity
oforigin. Aftertendays,hewastransferred toatertiary
hospitalforintractablediarrheaandmetabolicacidosis.
Uponadmissiontothetertiaryhospital,weighting2860g,
he presented generalized muscle atrophy, sparse
Figure 1 Simple abdominal radiography of a 2-month-old patient with total colon aganglionosis. Preoperative image shows dilated loops of the small bowel and colon, and low volumeofairinthecolonregion.
percussion,without visceromegaly.Anus ina normal posi-tionat inspection and, at the digital rectal examination, normotonicanalsphincterandrectalampullaofan appro-priatesizewereobserved.Thepatientpresentedexplosive dischargeoflargeamountsofliquidstooltothetouch.The diagnostichypothesisofHirschsprung’sdiseasewasraised, and anorectal manometry was performed. At that exam, itwasnot possible toregister rectoanal inhibitory reflex. An exploratory laparotomy was then performed; a transi-tionzonewasvisualized10cmbelowtheileocecalvalve.A totalcolectomywasperformed,including2cmofileum, fol-lowedbyileostomy.Ahistopathologicexamoftheresected segmentshowed complete absence of neuronal bodies in colonicsegmentsandscarceneuronalbodiesinthe myen-tericplexusoftheproximalilealsegmentattachedtothe stoma.
In the post-operative period,the patientevolved with intolerance(abdominaldistensionandbilious vomiting)to minimumvolumes of elemental formulainfused via naso-gastrictube.Asecondsurgerywasperformed,inwhichthe presence of bridle or kinking of the loops wasdiscarded; 10cmofterminalileumwereresected,thefixationofthe looptothestomawasre-made,andterminalileumbiopsies wereperformeduptothetransition withthe jejunum.In thesesamples,ganglioncellswereidentified;their morphol-ogyandnumberwere appropriateinthe ganglionicplexi. Inaddition,sampleswerefixedinformalinfor 48h, dehy-dratedin alcohol, andembedded in paraffin. Histological sectionsof 4m thicknesswere stained withhematoxylin
andeosinstainingforimmunohistochemicalstudyof inter-stitialcellsofCajal(ICC).Afterheatingandincubationwith peroxidase,thesectionswere incubatedwithanti-CD117
antibody, deparaffinized, placed in contact with specific antibodies,andstained.Forinternalcontrol, immunohisto-chemicalstainingkitsformastcellsinhumanneurofibroma wereused.ICCwerecountedintenmicroscopichighpower fields(HPF)(400×)inthevicinityofthemyentericplexus.
ThemeannumberofICCinsurgicalspecimensvariedwidely among the observed sections, with no relation with the height in relation to the ileostomy (median: 1.2; range: 0---9.7cells/HPF).Nodifferenceswereobservedinthe val-ues for each portion compared using the Mann---Whitney test (p=0.235), but when comparedwith values observed inhealthyadultstudies,thesevalueswerereduced.6
At12monthsofage,thepatientpersistsinneedof
par-enteralnutrition.
Discussion
In90%ofpatients,HDmanifestsitselfintheneonatalperiod, characterizedbydelayinthefirstpassageofmeconiumand progressiveabdominaldistension4;biliousvomitingoccurin
19---37%ofcases.7Thedelayineliminationofmeconiumin
thefirst24hoflifeisthestrongestindicatorofthecondition
andisreportedinupto90%ofpatientswiththedisease.7
However,somereportshavedemonstratedthatupto40%of
patientscaneliminatemeconiuminthefirsthoursoflife,
whichindicatesthatthisisnotarequiredsignfordiagnosis.8
The clinical features of HDTCA generally differ from
classicforms,namelytheincidencebetweensexes:inthe
classic form,itis fourmales toonefemale; inHDTCA, it
is 1:1. The clinical presentation with neonatal intestinal
obstructionmaynotbepartoftheclinicalpictureofHDACT.
Lateonsetofsymptoms(in thefirstweeksoflife oreven
later, assome cases arediagnosed in adolescence) is not
uncommoninthisformofthedisease.9InHDACT,microcolon
visualizationonbariumenemaisexpected;nonetheless,the
colonic loops sometimes present normal size without any
evidence of obstruction.10 Approximately 20% of patients
withHDTCA undergoa second distal resectionwith
surgi-cal repositioning of the distal loop at the stoma, as the
transition zone is irregularlyarranged and difficult to be
establishedintraoperatively.3Finally, 6.5%of patientswill
undergoconversionintopermanentileostomydueto
persis-tent motor dysfunction.5 In thecase reportedherein, the
loopdistensionobservedatprenatalultrasoundcouldhave
anticipated the diagnosis of intestinal obstruction, which
manifested lessintenselyin thefirstdaysof life,perhaps
duetoprotectiveeffectofexclusivebreastfeedingandits
recognizedprokineticactionandimmuneprotection.
The occurrence of severe diarrhea in neonates is an
exceptionalconditioninthosewhoareexclusivelybreastfed
and mayrepresent evidenceof serious complicationfrom
HD or another underlying disease. The enterocolitis
asso-ciatedwithHD is observedin 50%of cases,witha higher
frequency atthefirsttrimester andfirstyearof life,may
occurpriortooraftersurgery.7Thepreoperativeoccurrence
ismorecommonwhenthediagnosisismadeafterthefirst
monthoflife.7Thecomplicationmanifeststhrough
abdom-inaldistentionandexplosivediarrhea,whichprogressesto
vomiting, fever, lethargy, and shock. The diarrhea is not
causedbyaspecificintestinalinfection,butbythe
arelationshipbetweenthedysfunctionoftheenteric
ner-vous system, abnormal production of mucin, insufficient
immunoglobulinsecretion,intestinalmicrofloraunbalance,
andischemicmucosa.Therapyinvolveselectrolytic
recov-ery,colonicdecompression,andantibiotics.11
The postoperative periodinHDTCAis characterizedby
highmorbidity,includingfoodintolerance,electrolyte
dis-turbances, anddehydration due toexacerbated intestinal
secretion.Takingintoaccountthesecomplicationspriorto
surgery decreases mortality, but in the long run, thereis
greaterriskofstomapermanenceforlongperiodsinthese
patients.12Inthepresentcase,theanalysisoftheproximal
segment indicated hypoganglionosis, characteristic of the
transitionzone, whichmay explaintheneed for asecond
intervention.
Molecular biology and pathology anatomy techniques
in samples from patients with intestinal neuropathies
indicate that inflammatory and degenerative aggressions
resultingfromischemia,luminalstasis,andchangesinthe
microbiotamaycompromisethemorphofunctionalintegrity
of the enteric nervous system and of other elements
responsible for the intestinal motility, and may result in
postoperativemorbimortality.4
Quantitativechangesorthenetworkconnectionsofthe
ICChave been describedin variousconditionsthat evolve
withgastrointestinaldysmotility,suchasesophageal
achala-sia,gastroschisis,intestinalpseudo-obstruction,necrotizing
enteritis, inflammatory bowel disease, and anorectal
malformation.13
TheICCaredevelopedfromembryonicmesodermandare
locatedbetweenthenervefibersandsmoothmusclecellsof
theintestinalwall.Thesecellsarecapableoftransmitting
impulsesfromtheentericmotorneuronsandofgenerating
rhythmicelectricalslowwaveactivity.Theycoordinatethe
pacemakeractivityandpropagationofslowwaves,andact
inthesensitiveidentificationofmusclestretchphenomena.
Inthegastrointestinaltract,theyaredistributedfromthe
upperesophagealsphinctertotheinternalanalsphincter,13
juxtaposed tothenerve endingsof myentericmotor
neu-rons.The presenceof thetyrosine kinasereceptoronthe
cellmembraneallowedthedevelopmentofanti-c-kit
anti-bodies for the histological identification of these cells.14
TheabsenceorreductioninthenumberofICCdetermines
slow-waveabnormalities,causesdecreasedcontractilityof
the smooth muscle cells, and decreases the transit rate
oftheintestinalcontent.15
IntheaganglionicintestineandintheHDtransitionzone,
theremaybeareducednumberand/ordisturbancesinthe
communicationnetworkofc-kitpositiveICC.16Fewstudies
haveassessedtherelationshipbetweenHDandthe
distribu-tionofICCintheremainingganglionicsegment.Onestudy
of these cells in the remaining intestinal ganglionic
seg-ment of 15 children identified normal count in 13 cases,
which have evolved without postoperative dysmotility. In
twocases,however,the histopathologicstudy indicated a
significant reduction in the ICC count, and the patients
developed severe and persistent constipation,
necessitat-ingfurtherresectionoftheremainingdilatedsegment.17In
anotherseriesofcases,theICCcountinthecolonganglionic
11childrenwithHDwascomparedwiththecountinthe
con-trolgroup.Onepatienthadareducedcountanddeveloped
severeconstipation.18GfroererandRollereviewedthecase
reportsavailableandconcludedthatthereisconsiderable
heterogeneity in the ICC expression in the gut-associated
lymphoidtissueofpatientswithHD,andthatthedifferent
patternsofexpressioncouldresultfromdifferencesinthe
sizeofaganglionicsegmentstoaganglionicsegment
resec-tion,aswellasfromdifferencesincell-countingmethods.19
Thesefindingsrequirecarefulinterpretation,giventhe
scarcityofstudiesthatassociateICCcountandmotor
func-tioninhumans.Furthermore,theexaminedtissuesamples
werefrompatients withobstructivediseaseduring
differ-entperiods,whichmakesitdifficulttodeterminewhether
thereductionisaconsequence of theobstructionprocess
orapathogeneticassociationwithHD.Thediversityofthe
ICCdefectsmayindicatethatthisphenomenonissecondary
tochangesresultingfromintestinalobstruction,astheICC
areparticularlysensitive toischemia.14,15 The inability to
visualizeallcellsfromformalin-fixedparaffinsamplesisan
additionalfactorthathindersthedefinitionofa
histopatho-logicaldiagnosis.19
Inthepresentpatient,itwasnotpossibletocomparethe
histologicalsamplewiththoseofhealthychildren,ascarcely
available material.20 Appropriate tissue samples, correct
handling,andexpertiseinanatomopathological
interpreta-tionarecrucialtoincreasetheaccuracyofdiagnosisoflow
ICCcount.Itwouldbeworthwhiletoassessalargernumber
ofpatientswithunfavorableoutcomestodeterminethe
sig-nificanceofthehistopathologicalfindingofdecreasedICC
anditscorrelationwiththeclinicpresentation.
Funding
Thisstudydidnotreceivefunding.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.Amiel J, Sproat-Emison E, Garcia-Barcelo M, Lantieri F, BurzynskiG,BorregoS,etal.Hirschsprung’sdisease,associated syndromesandgenetics:areview.JMedGenet.2008;45:1---14.
2.BurkardtDD, GrahamJM Jr,ShortSS,FrykmanPK.Advances inHirschsprung’sdiseasegeneticsandtreatmentstrategies:an updatefortheprimarycarepediatrician.ClinPediatr(Phila). 2014;53:71---81.
3.Rabah R. Total colonic aganglionosis: case report, practi-cal diagnostic approach and pitfalls. Arch Pathol Lab Med. 2010;134:1467---73.
4.Moore SW.Totalcolonicaganglionosis andHirschsprung’s dis-ease:areview.PediatrSurgInt.2015;31:1---9.
5.LaughlinDM,FriedmacherF,PuriP.Totalcolonic agangliono-sis:asystematicreviewandmeta-analysisoflong-termclinical outcome.PediatrSurgInt.2012;28:773---9.
6.Alonso Araujo SE, Dumarco RB, Rawet V, Seid VE, BocchiniSF,NahasSC,etal.Reducedpopulationofinterstitial cellsofCajalinChagasicmegacolon.Hepatogastroenterology. 2012;59:2147---50.
8.Singh SJ, Croaker GD, Manglick P, Wong CL, Athanasakos H, Elliott E, et al. Hirschsprung’s disease: the Australian Paediatric Surveillance Unit’s experience. Pediatr Surg Int. 2003;19:247---50.
9.Lall A, Agarwala S, Bhatnagar V, Gupta AK, MitraDK. Total colonicaganglionosis:diagnosisandmanagementina 12-year-oldboy.JPediatrSurg.1999;34:1413---4.
10.Solari V,Piotrowska AP, Puri P.Histopathological differences betweenrecto-sigmoidHirschsprung’sdiseaseandtotalcolonic aganglionosis.PediatrSurgInt.2003;19:349---54.
11.Pierre JF, Barlow-Anacker AJ, Erickson CS, Heneghan AF, LeversonGE,DowdSE,etal.Intestinaldysbiosisandbacterial enteroinvasioninamurinemodelofHirschsprung’sdisease.J PediatrSurg.2014;49:1242---51.
12.Blackburn S, Corbett P, Griffiths DM, Burge D, Beattie RM, StantonM.Totalcolonicaganglionosis:a15-yearsinglecenter experience.EurJPediatrSurg.2014;24:488---91.
13.Al-Shboul OA. The importance of interstitial cells of Cajal inthegastrointestinal tract.SaudiJGastroenterol. 2013;19: 3---15.
14.Suzuki S, Suzuki H, Horiguchi K, Tsugawa H, Matsuzaki J, TakagiT,etal.Delayedgastricemptyinganddisruptionofthe
interstitialcellsofCajalnetworkaftergastricischaemiaand reperfusion.NeurogastroenterolMotil.2010;22:585---93.
15.BurnsAJ.DisordersofinterstitialcellsofCajal.JPediatr Gas-troenterolNutr.2007;45:S103---6.
16.MostafaRM,MoustafaYM,HamdyH.InterstitialcellsofCajal, the Maestro in health and disease. World J Gastroenterol. 2010;16:3239---48.
17.TaguchiT,SuitaS,MasumotoK,NagasakiA.Anabnormal distri-butionofC-kitpositivecellsinthenormoganglionicsegmentcan predictapoorclinicaloutcomeinpatientswithHirschsprung’s disease.EurJPediatrSurg.2005;15:153---8.
18.BetolliM,DeCarliC,Jolin-DahelK,BaileyK,KhanHF,Sweeney B, et al. Colonic dysmotility in postsurgical patients with Hirschsprung’sdisease.Potentialsignificanceofabnormalities intheinterstitialcellsofCajalandtheentericnervoussystem. JPediatrSurg.2008;43:1433---8.
19.GfroererS, Rolle U. Interstitialcells of Cajalin thenormal human gut and in Hirschsprung’s disease. Pediatr Surg Int. 2013;29:889---97.