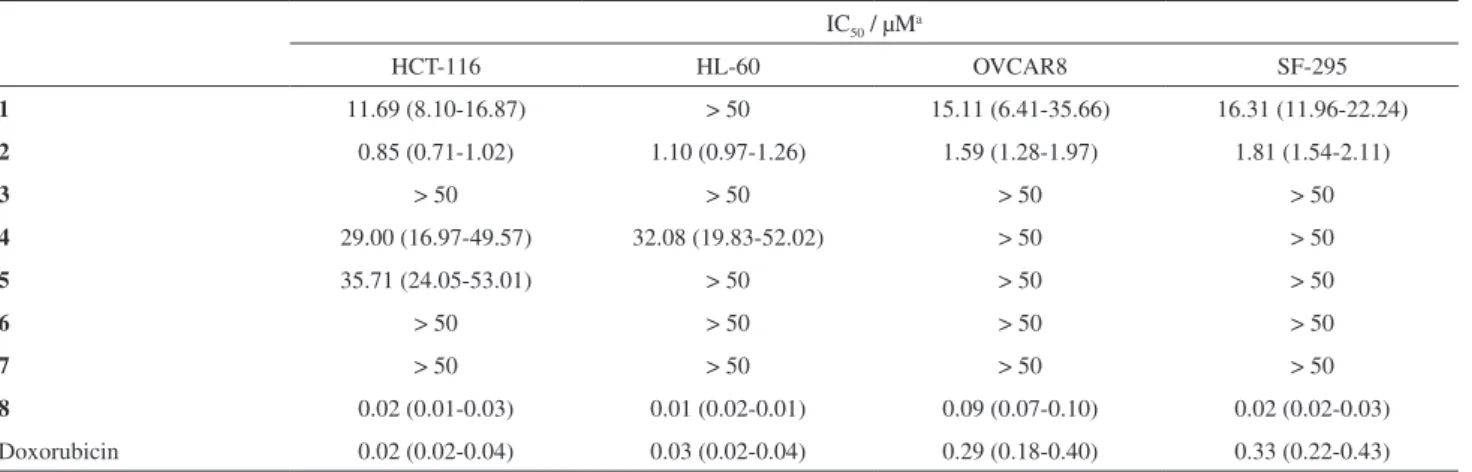
J. Braz. Chem. Soc. vol.26 número10
Texto
Imagem

Documentos relacionados
In this sense, the present work reports the development of an efficient and stable sensor based on CNT functionalized with the compound tris(1,10-phenantroline)copper(II)
A simple and practical pre-concentration method, dispersive liquid-liquid microextraction based on solidification of a floating organic drop (DLLME-SFO) technique, was proposed for
Analyzing the response surfaces of Figures 1, 2 and 3 it is possible to verify that the optimum conditions for extraction of compounds with antioxidant activity, as well as the total
This paper describes a sequential injection analysis method, with detection by square wave voltammetry using a hanging mercury drop electrode, to determine the herbicide picloram in
This work describes a simple and cost-effective method for quantitative determination of hydrochlorothiazide using biomimetic sensor based on a carbon paste modified with hemin
In this work, a method for multiclass determination of 70 pesticides residues with different properties in drinking water using solid phase extraction (SPE) with polymeric sorbent and
Three strains of Sphaerocavum brasiliense (CCIBt 3094, CCIBt 3096 and CCIBt 3316) isolated from different reservoirs in São Paulo State, and one (CCIBt 3179) from Rio Grande
test reactor) and to evaluate, separately, the total quantity of antioxidant compounds present in both hydrophilic and lipophilic emulsions phases using ORAC assay (oxygen