J. Braz. Chem. Soc. vol.26 número10
Texto
Imagem



Documentos relacionados
This study investigates the influence of the adhesive (epoxy resin) thickness and the dispersions of non-functionalized carbon based nanostructures (carbon nanotubes - CNT)
In spite of the fact that they have adequate germination results, they performed differently in field: good performance in field (GG-GPF), eight batches performed well in
NIR spectrometric determination of methane, ethane and propane was built by using quaternary mixtures of these gases with nitrogen, which were prepared by the automatic
A simple and practical pre-concentration method, dispersive liquid-liquid microextraction based on solidification of a floating organic drop (DLLME-SFO) technique, was proposed for
Analyzing the response surfaces of Figures 1, 2 and 3 it is possible to verify that the optimum conditions for extraction of compounds with antioxidant activity, as well as the total
This paper describes a sequential injection analysis method, with detection by square wave voltammetry using a hanging mercury drop electrode, to determine the herbicide picloram in
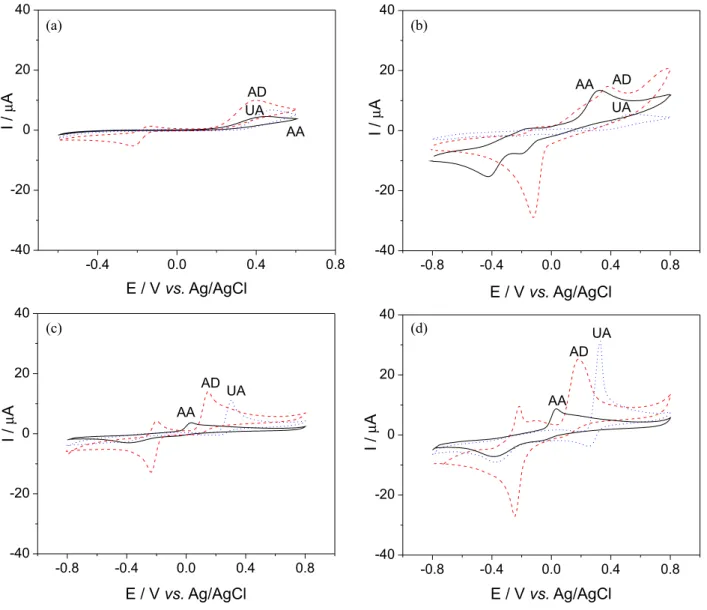
This work describes a simple and cost-effective method for quantitative determination of hydrochlorothiazide using biomimetic sensor based on a carbon paste modified with hemin
This paper reports the preparation and characterization of an organic-inorganic hybrid material for the removal of copper(II) from aqueous solutions, based on PDMS as an
