rev bras hematol hemoter. 2016;38(2):166–169
w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Case
Report
Bilateral
breast
plasmacytoma:
a
clinical
case
report
Thais
Rodrigues
da
Cunha
Fischer
∗,
Fabiana
Higashi,
Edvan
de
Queiroz
Crusoe,
Vania
Tietsche
de
Moraes
Hungria
FaculdadedeCiênciasMédicasdaSantaCasadeSãoPaulo(FCMSCSP),SãoPaulo,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory: Received3March2016 Accepted6March2016 Availableonline25March2016
Introduction
Theneoplasmofplasmacellsisagroupofdiseases encom-passing from monoclonal gammopathy of undetermined significance(MGUS)tomultiplemyeloma(MM).1
Conglomeratesofmonoclonalplasmacellsarecalled plas-macytomasandareincludedinthisgroup.2Theycanoccur
inisolationorassociatedwithMM,withthemostcommon locationbeingbonetissue.3
Theexistenceofextramedullaryplasmacytomashasalso beendescribed.Mostcommonly,theyaffectthehead,neck andsubcutaneoustissues,especiallyoftheupperrespiratory tract.4
Theyarepresentin7–19%ofpatientsatthetimeMMis diagnosed and in another 6–20% during the course ofthe disease.5
Breastplasmacytoma isarare condition.In2006,Taylor etal.,2described43casesintheliterature.Diagnosisismade
bypathologyhoweverimagingcanfurtheraidthediagnosis ofthedisease.Ultrasoundandmammographicaspectshave
∗ Correspondingauthorat:RuaCapitãoMacedo,314/44,VilaMariana,04021020SãoPaulo,SP,Brazil.
E-mailaddress:[email protected](T.R.d.C.Fischer).
already beendescribed intheliterature,ashaveaspectsof magneticresonanceimaging.2–4AcaseofMMwithmammary
glandinvolvementatdiagnosisisdescribedherein.
Case
report
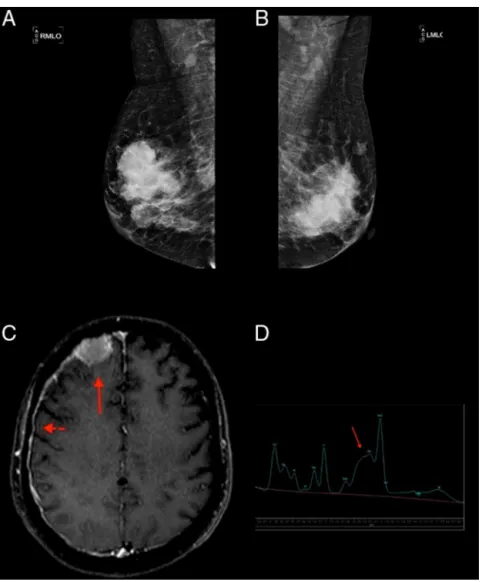
A 52-year-old, Black female patient was admitted to hos-pital withasthenia. She had suffered 10kg ofweight loss, paininthethoracicregionandvomiting.Thephysical exam-ination showed pain on palpating the chest and bilateral breast lumps.Initialtestsshowedbicytopenia (anemiaand thrombocytopenia), renal dysfunction, and hypercalcemia. First, the symptomatic hypercalcemia was controlled and, subsequently,aninvestigationinto theetiologywascarried out, with mammary neoplasm being the initial hypothe-sis.Mammographyshowedhyperdensenodules,withregular marginsandlobulatedcontours,distributedbilaterally(Breast Imaging-Reportingand Data System –BI-RADS0), whereas ultrasonographyshowedmultiplesolidoval-shapednodules with lobulated contours and with vascularization by color
http://dx.doi.org/10.1016/j.bjhh.2016.03.003
rev bras hematol hemoter. 2016;38(2):166–169
167
Figure1–(AandB)Mammography:hyperdenseandlobulatednoduleswithcircumscribedmargins;(C)extra-axial expansivelesioncompatiblewithplasmocytomaintherightfrontalconvexity(arrow)withpachymeningealimpregnation (arrowhead),demonstratingcentralnervoussysteminvolvement;(D)spectroscopyofthesamepatientshowingan increasedglutamine–glutamatecomplex(arrow)resultingfromelevatedcentralnervoussystemammonia.
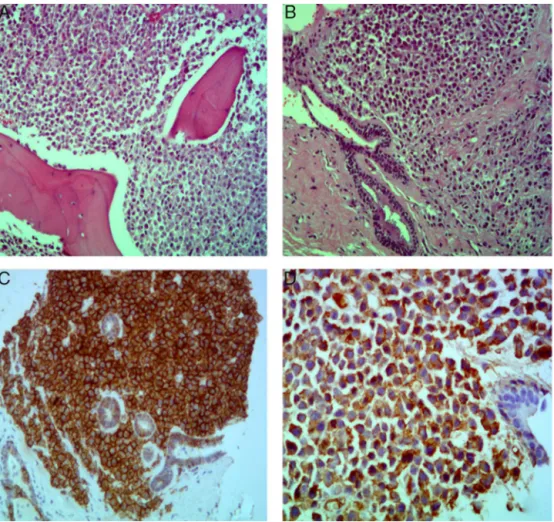
Doppler(Figure1).Acorebiopsywasperformed.Testresults, however, ruled out primary breast cancer while showing breastplasmacytoma,withlambdachainrestriction,plasma cell ratio of95%,CD138+ and negative forCD56(Figure 2). OthertestsconductedtoconfirmMMwerepositive.Protein electrophoresisidentifiedmonoclonalcomponents(5.34g/dL), immunofixation identified IgA/Lambda and bone marrow biopsyshowedbonemarrowplasmacytosis90%,CD138+,and Lambda+ (Figure 2). Hence, a diagnosis ofIgA/lambda MM withextramedullaryplasmacytomalocatedinthebreastwas reached,withaninternationalstagingsystemscoreofIIIand DurieSalmon(DS)stageofIIIB.
The patient underwent induction chemotherapy with theadministrationoftheCTDregimen(cyclophosphamide, thalidomide,anddexamethasone).Afterthreecycles,her clin-icalconditionworsenedwithalteredlevelofconsciousness, seizures, and disease progression with new plasmacy-tomas (sternum and central nervous system−CNS) and
hyperammonemia being observed. This metabolic change
was established by measuring the serum ammonia level (93mol/L; normal value up to 32mol/L) and confirmed by proton magneticresonance spectroscopy (PMRS), which showedaglutamine/glutamatepeakandreducedcholineand myo-inositol(Figure1).
The chemotherapy regimen was then changed to a combination of dexamethasone, thalidomide, doxorubicin, cisplatin,cyclophosphamide,andetoposide(DT-PACE).After three cycles of this chemotherapeutic regimen, the mono-colonal component reduced completely and there was a reductionintheextramedullaryplasmacytomas(breast, ster-num, and CNS).Treatment then proceededwithhigh-dose melphalanandrescuewithautologoushematopoietic progen-itorstem celltransplantation and subsequentradiotherapy againstthesternalplasmocytoma.
168
rev bras hematol hemoter. 2016;38(2):166–169Figure2–(A)Breastand(B)bonemarrowbiopsies,bothwithhematoxylinandeosin(HE)stainingcharacterizedby
infiltrationofplasmacells;Breastbiopsyimmunochemistrywith(C)lambdarestrictionand(D)CD138+immunoexpression.
intrathecal chemotherapy (methotrexate, cytarabine, and dexamethasone).
She evolved to death ten months after diagnosis, with multiple extramedullary plasmacytomas (breast, sternum, CNS,subcutaneoustissue)and absence ofany monoclonal component.
Discussion
Extramedullary plasmacytomasofthe breast are rare;they havebeenfoundatdiagnosisinonly14%ofcases.Most(75%) plasmacytomasofthebreastoccurduringrelapseofMM.2
Theepidemiologicalcharacteristicsofthecasedescribed hereinareconsistentwiththedataintheliterature,regarding genderandage.Mostbreastplasmocytomasoccurinwomen withameanageof53yearsatdiagnosis,butpredominantly unilateral.2,3
Themammographicresultsofthis patientare in agree-ment with the findings reported in the literature as well-defined masses with irregular margins. On the other hand,theultrasonographic patterndescribed byauthors is heterogeneous, with hyperechoic, hypoechoic, or anechoic lesionsbeingreported.3
The prognosis of patients with extramedullary plasma-cytoma is variable. Solitary plasmacytomas have a good prognosis (radiotherapyand surgeryarecurative treatment options)andlessthan30%ofcasesdevelopMMorother plas-macytomas,whereasthediseaseprogresseswithunfavorable outcomesinpatientswithMMandextramedullaryprimaryor secondaryplasmacytomas,asinthiscase.5
Progressionofthediseasewasobservedinthecurrentcase withnewextramedullaryplasmacytomasdespiteoftheCTD regimen. Using PMRSand determinationof serum ammo-nialevels,hyperammonemicencephalopathywasalsofound. ThisconditiontogetherwithMMandwithoutliverdiseaseis rare(3.8%).Nevertheless,itshouldbeconsideredinpatients with altered levels of consciousness, as observed in this patient.6,7
Hyperammonemiawasnotconsideredastheonlysingle factor forthe alteredmental status,but part ofacomplex scenario, which included hyperviscosity of the blood, CNS infiltration,andhypercalcemia.
Thisencephalopathyiscausedbytheproductionof ammo-niaatcriticallevelsbytheplasmacellsthemselves,thereby exceedingthedepurationcapacityofhepatocytes.8
InthesystematicreviewbyPhametal.,6
rev bras hematol hemoter. 2016;38(2):166–169
169
subtypesIgAandIgG,advancedstagesofthedisease,andCNS infiltration,whichleadstoaworseprognosiswithhigherrates ofhospitalizationandmortality.9,10
Considering the progression and aggressiveness of the disease,thechoicewastomodifytreatmenttotheDT-PACE regimen.11 Bladè et al.12 found that extramedullary
dis-ease appears to be more properly treated with intensive chemotherapy regimens, including new agents such as lenalidomide and pomalidomide, compared to traditional therapiesforMM;however,thisoptionisnotclearlydescribed intheliteratureyet.
Extramedullarydisease,whetherthepatienthas plasma-cytomasoftheboneornot,maybethemostimportantclinical findinginthecourseofMM,asinthis case.Thebiological mechanismsunderlyingthisoccurrencehavenotbeen prop-erlyestablished,butaseriesofeventsarepostulated.These includingdecreasedexpressionofadhesionmolecules(CD44 andCD56),increasedangiogenesis,lossofcytokinereceptors responsibleforplasmacellhoming[C-X-Cchemokine recep-tortype4(CXCR4)andC-X-Cmotifchemokine12(CXCL12)] andbonemarrowhypoxemia,causingthesecellstomigrateto theperiphery.AlthoughthelossofCD56expressionfacilitates hematogenousorbonedissemination,itsexpressionis para-doxicallyassociatedwithimpairmentofspecificsites,suchas
theCNS.13,14
The heterogeneity in CD56 marking could be duly evidencedinthiscase,wheretherewaslossofCD56 expres-sion in the plasmocytoma of the breast and, during the patient’sprogression,thismarkerwasshowntobepositive whenimmunophenotypingtheliquor;thissuggeststhe influ-enceofasyetunknownmechanismsandotherfactors.
Althoughcytogenetics assays were not carriedout, this does not seem to be a finding capable of predicting out-comes.Vargaetal.,15inastudyof117patients,demonstrated
that unfavorable cytogenetics results at diagnosis did not determinetheoccurrenceofextramedullarydiseaseorpoor prognosis.
Theclinicalcharacteristics andlaboratoryresultsofthis patient were already indicative of a poor prognosis with lowchances ofsurvivalatdiagnosis (ISS: III). Her progres-sion with extramedullary plasmocytomas, probably due to hematogenousdissemination,onlycorroboratedthe biolog-ical complexity of the disease. The median survival time reportedforthesecasesistenmonthsinspiteofthetreatment administered,aswasseeninthiscase.13,15
Conflict
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.HuberH,HohnMJ,RachelR,FuchsT,WimmerVC,StetterKO.
AnewphylumofArchaearepresentedbyananosized
hyperthermophilicsymbiont.Nature.2002;417(6884):63–7.
2.TaylorL,AzizM,KleinP,JagannathS,AxelrodD.
Plasmacytomainthebreastwithaxillarylymphnode
involvement:acasereport.ClinlBreastCancer.2006;7(1):81–4.
3.BremRF,RevelonG,WilleySC,GatewoodOM,ZeigerMA.
Bilateralplasmacytomaofthebreast:acasereport.BreastJ.
2002;8(6):393–5.
4.LamyO,vonBremenK,BurckhardtP.Breastplasmacytoma.
LeukLymphoma.2000;37(5-6):611–5.
5.UsmaniSZ,HeuckC,MitchellA,SzymonifkaJ,NairB,Hoering
A,etal.Extramedullarydiseaseportendspoorprognosisin
multiplemyelomaandisover-representedinhigh-risk
diseaseevenintheeraofnovelagents.Haematologica.
2012;97(11):1761–7.
6.PhamA,ReaganJL,CastilloJJ.Multiplemyeloma-induced
hyperammonemicencephalopathy:anentityassociatedwith
highin-patientmortality.LeukRes.2013;37(10):1229–32.
7.TalamoG,CavalloF,ZangariM,BarlogieB,LeeCK,
Pineda-RomanM,etal.Hyperammonemiaand
encephalopathyinpatientswithmultiplemyeloma.AmJ
Hematol.2007;82(6):414–5.
8.OtsukiT,YamadaO,SakaguchiH,IchikiT,KouguchiK,Wada
H,etal.Invitroexcessammoniaproductioninhuman
myelomacelllines.Leukemia.1998;12(7):1149–58.
9.FassasAB,MuwallaF,BerrymanT,BenramdaneR,JosephL,
AnaissieE,etal.Myelomaofthecentralnervoussystem:
associationwithhigh-riskchromosomalabnormalities,
plasmablasticmorphologyandextramedullary
manifestations.BritJHaematol.2002;117(1):103–8.
10.KyleRA,GertzMA,WitzigTE,LustJA,LacyMQ,DispenzieriA,
etal.Reviewof1027patientswithnewlydiagnosedmultiple
myeloma.MayoClinProc.2003;78(1):21–33.
11.LeeCK,BarlogieB,MunshiN,ZangariM,FassasA,JacobsonJ,
etal.DTPACE:aneffective,novelcombinationchemotherapy
withthalidomideforpreviouslytreatedpatientswith
myeloma.JClinOncol.2003;21(15):2732–9.
12.BladéJ,FernandezdeLarreaC,RosinolL.Extramedullary
diseaseinmultiplemyelomaintheeraofnovelagents.BritJ
Haematol.2015;169(6):763–5.
13.BladéJ,FernándezdeLarreaC,Rosi ˜nolL,CibeiraMT,Jiménez
R,PowlesR.Soft-tissueplasmacytomasinmultiplemyeloma:
incidence,mechanismsofextramedullaryspread,and
treatmentapproach.JClinOncol.2011;29(28):3805–12.
14.GangatharanSA,CarneyDA,PrinceHM,WolfMM,
JanuszewiczEH,RitchieDS,etal.Emergenceofcentral
nervoussystemmyelomaintheeraofnovelagents.Hematol
Oncol.2012;30(4):170–4.
15.VargaC,XieW,Laubachj,GhobrialIM,O’DonnellEK,
WeinstockM,etal.Developmentofextramedullarymyeloma
intheeraofnovelagents:noevidenceofincreasedriskwith
lenalidomide–bortezomibcombinations.BritJHaematol.