Loal Order Struture of a-SiO
x N
y
:H Grown by PECVD
W.L. Sopel, M.C.A. Fantini,
InstitutodeFsia,USP
M.I. Alayo and I. Pereyra
Departamento deEngenhariadeSistemas Eletr^onios,EPUSP
13.081-970S~aoPaulo,SP,Brazil
Reeivedon23April,2001
Inthis workwestudythestrutural propertiesofamorphous oxynitridelms(a-SiOxNy),grown
byplasma enhaned hemial vapour deposition(PECVD)at320 0
C.Thelmsweredeposited at
dierent owratio ofN2OandSiH4. Theatomiompositionofthe sampleswasdeterminedby
meansof Rutherfordbaksattering spetrometry (RBS). The loal order struture was studied
byX-ray absorptionspetrosopy (XAS)andthe hemial bondingswereinvestigatedby Fourier
transforminfrared (FTIR) spetrosopy. The results evidene a tetrahedri arrangement of the
oxynitridenetwork. ThetetrahedronsaresimiliartoSiO3Nforxvaluesbetween1.43and1.64. For
xvalueshigherthan1.64thetetrahedronsaresimilartoSiO4.
ThePlasmaEnhanedChemialVapourDeposition
(PECVD) methodhasbeenextensivelyapplied to
ob-tainsilionbaseddieletrilms. Inpartiular,theuse
of SiO
2
and SiO
x N
y
lms, grown by PECVD at low
temperature, beameveryattrativedue to the
possi-bilityofintegratingoptialandeletrialdeviesinthe
samehip[1,2,3℄. Moreover,PECVDisauseful
teh-nique to deposit thik lms, about 4-5 (m), a
om-mon requirement in optial devie tehnologies. The
fundamental advantageof this tehnique is the
possi-bility of ontrollingthe hemial omposition, as well
as the strutural and optial properties of the silion
oxynitridelms,byadequatelyadjustingthedeposition
parameters[4,5,6℄. Ourpreviousworks[4,7,8,9℄
estab-lishedtheexperimentalonditionstogrowa-SiO
x N
y :H
lmsathighdepositionrates,havingaontinuous
vari-ation in itsrefrativeindex, withontrolled thikness,
seletedhemialbondsand improvedmorphology. In
partiular, high quality, low temperature a-SiO
2 lms
were deposited.
The aim of this work was to determine the
lo-al atomi struture aroundthe Si atoms of our good
quality a-SiO
x N
y
:H lms asafuntion of their
hem-ialomposition. Theatomi ontent was determined
byRutherfordbaksatteringspetrometry(RBS);the
hemial and strutural properties were investigated
byextendedX-rayabsorptionnestruture(EXAFS),
X-ray absorption near edge struture (XANES) and
Fouriertransforminfrared spetrosopy(FTIR).
omposition, were performed at LAMFI/USP, S~ao
Paulo, using a He +
beam with energy E=1.7 MeV,
harge Q=30C, urrentI= 30nA anddetetion
an-gle=170 o
. TheRBSdatawereanalyzedthroughthe
RUMProutine[10℄andprovidedtheSi,NandOatomi
density(atoms.m 2
).
Theanalysis of thehemialbondswasperformed
by means of the Fourier transform infrared (FTIR)
spetrosopy in a BioRad FTS-40 spetrometer, on
lmsdeposited overpolyrystallinesilion wafer,with
highresistivity. Thelmsweremeasuredatroom
tem-perature in a nitrogen atmosphere, with a resolution
of 4m 1
. TheH ontentin the lmswasalulated
using theFTIRN-H signal. Itdereases from5at. %
in the mostnitrogenated lm to zeroin lms without
detetable amountofH.
The XAS (X-ray absorption spetrosopy)
experi-mentswere arriedoutat theSXSbeamline[11℄of the
SynhrotronLightNationalLaboratory(LNLS,
Campi-nas,Brazil). Themeasurementswereperformedin the
energyrangeof(1800-2300)eV,usingadouble-rystal
InSb(111)monohromator. TheEXAFSspetraofthe
a-SiO
x N
y
lms,a-SiO
2
and -Si
3 N
4
referenesamples
werereordedat the SiK-edge, at roomtemperature,
beingolletedinTEYmode,with1eVstep. Further,
moreauratemeasurementsweredoneinthenear
ab-sorption edge (XANES)region, in the (1840-1870)eV
energyrange,with0.2eVstep.
onen-lyzedlms. It's learfromthistable thatthehemial
ompositionisdependentonthedepositiononditions.
Infat,the[O/Si℄onentrationratiovariedfrom1.43
to 1.85with theinreasing oftheN
2
O owin the
de-position hamber. On theother hand,the[N/Si℄
on-entrationratiodereasedalmost6times(from0.57to
0.12)asthenitrousoxideowinreased. It'simportant
topointoutthatthe[(O+N)/Si℄onentrationratiois
alwaysloseto2.
TableI.Samplename,owratio[N
2 O/SiH
4 ℄ and
onentrationatomiratiox=[O/Si℄andy=[N/Si℄
determinedbyRBStehnique.
Sample [N
2 O/SiH
4
℄ x y
A 2.0 1.43 0.57
B 2.5 1.57 0.34
C 3.0 1.64 0.29
D 4.0 1.80 0.12
E 5.0 1.85 0.12
The infrared spetra of the a-SiO
x N
y
lms are
shownin Fig. 1. It should be notedthat the 2500to
4000m 1
spetralregionwasmagnied5timesin
or-dertobetterappreiatetheN-Hband.TheFTIRdata
presentaontinuousshiftinthepeakpositionofthe
Si-Ostrehingvibrationmodetolowerfrequenies,asthe
amountofOinmaterialmatrixdereases. Thisshiftis
explainedbythepreseneoftheSi-Nbonds. Also,the
shoulder at around1250m 1
[12, 13℄, harateristis
of SiO
2
, beomes less dened. The 3400 m 1
band,
orrespondingto the strething vibrationmode of the
N-Hbonds,indiatesthepreseneofN-Hbondsinlms
A, B and C, that have higher nitrogen ontent. The
samplewithhighoxygenontent(sampleE)presentsa
FTIR spetrumverysimilar to the thermiallygrown
a-SiO
2 .
Figure1. FTIRspetraforallstudiedsamples,seetableI.
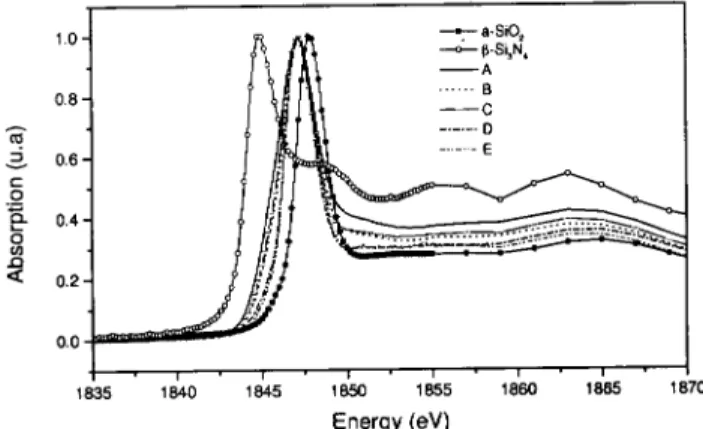
TheXANES spetraof amorphoussilion dioxide,
oxynitridelmshavebeentakenatthesilionKedge,
as shown in Fig. 2. The spetra of -Si
3 N
4
and
a-SiO
2
show no signiant dierene from those
previ-ouslyreported[14,15℄. FromFig. 2,oneanseestrong
thresholdpeaksofthea-SiO
x N
y
lms,loatedbetween
-Si
3 N
4
anda-SiO
2
,losesttoa-SiO
2
. Thereisabroad
struture between 1860 and 1870 eV, for all XANES
spetra,similiartoa-SiO
2 .
Figure2. ComparisonbetweemtheSiKedgeXANES
spe-trafora-SiO
2 ,-Si
3 N
4
andsamples(tableI).
The XANES spetra of all samples and of a-SiO
2
refereneareverysimilarandonrmstheamorphous
stateof our samples. One an notie the existene of
ahemial shiftfrom silion nitride to siliondioxide.
This shift indiates that the threshold position is
de-pendent on the nature of the atoms whih build the
tetrahedron environment of Si and is diretly related
to the eletronegativity of the surronding atoms. We
anobservethat the threshold position of all samples
hanges ontinuously but it stays between the values
ofthesilionnitride andsiliondioxidereferenes(see
Fig. 2). This result is onsistent with the formation
of afourfold oordinate Si network suh that Si is
ei-theronnetedtoOand/orNintetrahedrahavingthe
SiO
N
4
(=0,1,2,3and4)ongurations.
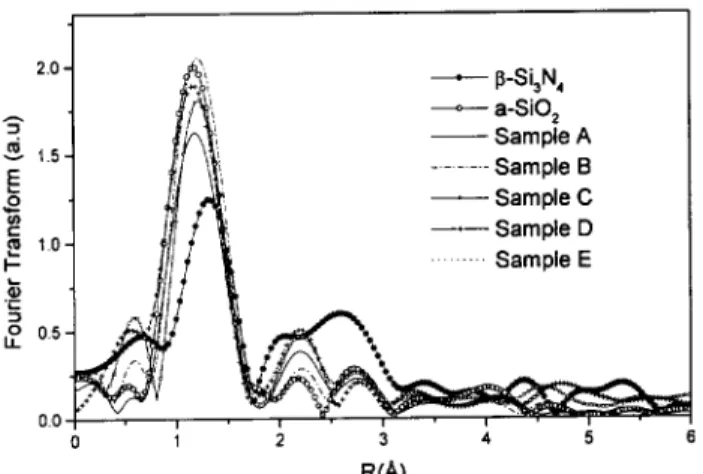
TheFouriertransform(F.T.)inthereal-spae(also
referredto R-spae)ofthek 2
-weightedosillationwas
alulatedoverthesameenergyrange(1.5-9.5
A)for
all samples and referenes. The bak Fourier
trans-forminthereiproal-spae(k-spae)orrespondingto
agivenoordinationshellwasttedwitheletroni
pa-rametersextrated from thereferene samples, whose
struturearesimilartothosebeinginvestigatedin this
work.
TheFouriertransformurvesobtainedforthe
sam-ples (Fig. 3), an give us some qualitative
informa-tion about the loal order struture. First, one an
observe the absene of long-range order (for R values
higherthan3
A). Nevertheless,theshortrangeorderis
slightlydependentonthehemialomposition,as
Figure 3. F.T. of the a-SiO2 and -Si3N4 referenes and
samples(tableI).
The EXAFS data analysis was done onsidering
a two-shell model with partial oordination numbers
N
N O andN
Si O
whihprovidesanaveragepitureof
theloalarrangementoftheNandOatomsintherst
oordinationshellsurroundingtheabsorbingSiatoms.
TheEXAFS resultsprovide theoordinationnumber,
averageinteratomidistaneandtheDebye-Waller
fa-tors,aspresentedintableII.
Table II. First shell strutural parameters obtained
with two-shell t (Si-O + Si-N) for dierent
sam-ples (see table I), where N
i
(10%),
i
(15%) and
R
i
(1%) orrespond to oordination number,
Debye-Waller fator(
A 1
), and interatomi distane (
A),
re-spetively(i=1,Oxygen,i=2,nitrogen).
Sample N
1
1 R
1 N
2
2 R
2
A 2.8 0.036 1.60 1.3 0.001 1.70
B 3.1 0.000 1.63 1.2 0.000 1.64
C 3.2 0.000 1.63 0.8 0.000 1.66
D 4.0 0.000 1.60 - -
-E 4.0 0.000 1.60 - -
-The results show that the disorder of the lms is
omparableto that ofthereferene ompounds, being
largerforthelmwiththehighestnitrogenontent.
The RBSdata showedthat [(O+N)/Si℄
onentra-tionratioisalwaysloseto2. Thisresultsuggeststhat
bothoxygen andnitrogen atomsoupytherst
oor-dination shell aroundtheSi atoms, keepingthe
tetra-hedralSioordination. Thisstruturalmodelwas
on-rmedbythettingof theEXAFSsignal,onsidering
thattherstSioordinationshellispartiallylledwith
O and N atoms, whose onentration was taken from
theRBS data. These results, togetherwith theFTIR
data,suggestthatthebetterdesriptionofthematerial
isahomogeneousnetworkformed bySiO
N
4
tetha-Aknowledgments
Thisworkwassupported byFAPESP/Brazil
(pro-ess number: 98/09806-6). We aknowlegde Dr. F.C.
Vientin for the XAS measurements at LNLS/Brazil
andDr. M.H. TabaniksfortheRBSmeasurements.
Referenes
[1℄ U. Hilleringmann and K.Goser, Miroeletroni. Eng.
19,211(1992).
[2℄ M. Tabasky,E.S. Bulat, B. Tweed, and C.Herrik, J.
Va.Si.Tehnol.A12,1244(1994).
[3℄ A.Borghesi,A.Sassella,B.Piva,andL.Zanotti,Solid
StateCommun.100,657(1996).
[4℄ I. Pereyra, M.I. Alayo, J. Non-Cryst. Solids 212, 225
(1997).
[5℄ J. Viard, R. Berjoan, J.Durand, J. Eur. Ceram.So.
17,2001(1997).
[6℄ C.F. Lin, W.T.Tseng,M.S. Feng, Jpn.J.Appl. Phys.
Pt.137,6364(1998).
[7℄ M.I. Alayo and I. Pereyra, Braz. J. Physis 27A, 146
(1997).
[8℄ M.I. Alayo, I. Pereyra, and M.N. Carre~no, Thin solid
Films332,40(1998).
[9℄ W.L. Sopel,R.R. Cuzinatto,M.H.Tabaniks,M.C.A.
Fantini, M.IAlayo,andI.Pereyra,aeptedinJ.
Non-Cryst.Solids,(2001).
[10℄ L.R.Doolittle,Nul.Instrum.Meth.B 9,344(1995).
[11℄ M. Abbate, F.C. Vientin, V. Compagnon-Cailhol,
M.C. Roha, and H. Tolentino, J. Synhrotron
Radia-tion.6,964-972(1999).
[12℄ P.G. Pai, S.S. Chao, Y. Takagi, and G.Luovsky,J.
Va.Si.Tehnol.A4,689(1986).
[13℄ H.J. Shiliwinski, U. Shnakenberg, W. Kindbrake,
H. Ne, and P.Lange, J.Eletrohem. So. 139, 1730
(1992).
[14℄ J.Viard,E.Behe,J.Duran,andR.Berjoan,J.Eur.
Ceram.So.17,2039(1997).
[15℄ K.M.Behrens,E.D.Klinkenberg,J.Finster,andK.H.
Meiwes-Broer,Surf.Si.404,729(1998).
[16℄ K.v.Klitzing, G.Dorda, and M. Pepper, Phys.Rev.
Lett.45,494(1980).
[17℄ H. Ibah and H. Luth, Solid-State Physis,
(Sringer-Verlag,1993).
[18℄ K.vonKlitzing,unpublished.
[19℄ D.C.Tsui,H.Stoermer,andA.C.Gossard,Phys.Rev.
Lett.48,1559(1982).
[20℄ H. Stoermer, Pro. 2nd Braz. Shool of Semiond.
Phys., Eds. J.R. Leite andC.E.T. Gonalves daSilva,
p.539.