Preparation and Charaterization of Nanorystalline
h-BN Films Prepared by PECVD Method
J. Vilarromero
,M.N.P. Carre~no, I. Pereyra,
GNMD,PSI,EPUSP, UniversityofS~aoPaulo,
CEP0524-970, P.OBox61548, S~aoPaulo,Brazil
N. C. Cruz, and E.C. Rangel
LPA,DFQ,FauldadedeEngenharia, Unesp-Guaratingueta
CEP12516-410,S~aoPaulo,Brazil
Reeivedon23April,2001
This workdesribesasystematistudyofthepreparationof nano-rystallinethinh-BNlmsby
Plasma-EnhanedChemialVaporDeposition(PECVD)tehnique. Thesampleswerepreparedat
lowtemperaturesusingB2H6andN2 asgaspreursors. Itisshownthattheowratioamongthese
gaseshasanimportantinueneonthesize oftherystallites(deduedbyRamanspetrosopy).
The B2H6/N2 ow ratio was variedfrom 1,6 x 10 2
to 6.7 x 10 4
leading to lmspresenting a
rystallite size, whihvariedfrom90nmtoamorphous, respetively. TheXRDspetrashowtwo
peaksat2valuesaround42 o
and44 o
,whihareassoiatedto<110>and<100>h-BNrystalline
diretions, indiating,a <002 >preferential orientationfor theh-BN rystallites. The
thermo-mehanialproperties,asstress,hardness,andYoungmoduluswerealsostudiedandorrelatedwith
the struturalproperties. Theompositionofthelmswasobtainedby RBSandEDSindiating
BorontoNitrogenratio,losetostoihiometryforallthestudieddepositiononditions.
I Introdution
Inanalogytothearbonsystem,BNanexistin
dier-entrystallographiphases,amongthemthehexagonal
and the ubi modiations; h-BN and -BN have
re-eived great attention in view of oatingappliations.
Likegraphite h-BN onsists of sp 2
-bonded hexagons.
In h-BN, the boron and nitrogen atoms alternate in
sequene of hexagonal layers. Sub modiations with
higherdisorderinthelayersequenearepossible.
Nev-ertheless, the h-BN hasbeenonsidered asone ofthe
most promising ne eramis due to its high
eletri-al resistivity, exellent thermal ondutivity and
ex-tremelylowdieletri onstant[1-7℄. It has also been
developedasapotentialalternativeforberoatingsin
eramimatrixompositesonaountofitslubriation
andorrosion-resistantharateristis[7℄.
Dierent PVD (physial vapor deposition) and
CVD (hemial vapor deposition) deposition methods
toprepareBNhavebeenreported,normallyin
temper-aturesabove600 0
C[8-11℄. ToprepareBNatlow
tem-perature is advantageous, however depending on the
nal thikness the lms present strutural instability
related to internal stress. In this way the knowledge
ofthemehanialpropertiesofthesematerials,suhas
stress, hardness, elasti modulus, and thermal
expan-sion oeÆient is of great interest. It is well known
stronglyorrelated withthelm struturesuh as
de-fets,voids,networkstrainandthemeanoordination
number. Inthispaper,wepresentasystematistudyof
theinueneofthesubstratetemperatureanddiborane
tonitrogenowrateonthestruturalandthermo
me-hanialpropertiesofthehexagonalboronnitridethin
lmspreparedbyPECVDmethodatlowtemperatures
(below400 0
C).
II Experimental
The Boron Nitride thin lms were prepared using a
standard13.56 MHzradio frequeny PECVDreator,
apativelyoupled,andutilizingdierentmixturesof
(B
2 H
6 / N
2
) as gas preursors. The set-up has been
desribed in detail elsewhere [12, 13℄. The rf power,
substrate temperature,diboranetonitrogen owrate,
andthetotalpressureweresystematiallyvariedfrom
onedepositiontotheotherinordertoanalyzetheeet
oftheseparametersonthelmsproperties.
Rutherfordbaksattering(RBS),X-ray
photoele-tronspetrosopes(XPS),andEnergyDispersiveX-ray
(EDS) spetrosopyandElastiReoil detetanalyses
(ERDA) were used to determine the hemial
ompo-sition of the samples. In addition, XPS spetra were
obtainedbyFTIR(inaBio-Radspetrometer)inlms
deposited onto -Si substrates, in the 200-4000m 1
wave-number range. The miro-Raman spetra were
obtainedwithaRenishawspetrometerin
baksatter-ingonguration,atroomtemperatureusingthe
514.5-nm line of argon laser and X50 amplier mirosope.
XRD measurementswere performed inaPhilips
spe-trometer using a Cu soure in a low angle system to
onrmthenano-rystallinephase.
Stress measurements were taken from lms
de-positedonto3x25x0.4mm 3
(111)-SibarsusingaSloan
prolometertodeterminetheradiusofurvatureofthe
lm-substrate omposite. The stress was then
alu-lated bytheStoney equation[14℄:
=[ E=(1 ) ℄ t 2
=6dR
(1)
where E, and t are the Young's modulus, Poisson's
ratio, and thiknessof thesubstrate, R and d are the
radius of urvature and the thikness of the lm,
re-spetively. ThehardnessandYoungModuluswere
ob-tained in aNanohardness systemat dierent stepsof
measurements.
III Results
Theompositionalanalysesindiatethattheprodued
lmsarelosetostoihiometrywiththeB/Nratio
vary-ingfrom1to1.15dependingonsamplepreparationand
experimental onditions. Theonentrationofarbon,
oxygenandotherelementsfoundasontaminantswas
low but inrease for samples prepared with low
sub-stratetemperature. Bothmoleular hydrogenand
hy-drogenbondedtoboronandnitrogeninreasewhenthe
substratetemperaturesdereaseasobservedbyERDA
and infraredspetra,respetively. Thedeposition rate
variedfrom2x10 2
to3.3x10 1
A/sandthesample
thiknessfrom 0.35to 0.8m.
TheXPSspetraforh-BNthinlmsprepared
keep-ingallthedepositionparametersonstantandvarying
thesubstratetemperaturearepresentedinFig. 1. The
hexagonal phase of boron nitride ompound presents
the B1sorelevelat 190.5eV [15℄. All ourBN
sam-ples present a B 1s-ore level at 190.3 eV very lose
tothehexagonalBNphase. However,thesamples
pre-paredatroomtemperaturepresentalowshoulderband
at 192eV, whih is assoiated to boron oxide [15℄, in
aordaneto omposition analyses, whih indiates a
higher presene of oxygen, ompared to samples
pre-pared at 340 o
C. In Fig. 2, the infrared spetra for
thesamesamplespresentedatgure1areshown. The
absorption band observedaround 780 m 1
and 1380
m 1
are related to theBN hexagonalphase[16℄ and
theabsorption bandsenteredat 2450m 1
and 3250
m 1
arerelatedtohydrogenbondedto boronand
ni-thedepositiontemperatureshowsthatforhigher
tem-peraturesthehexagonalrystallitesize,asindiatedby
theFWHMoftheh-BNabsorptionbandat1380m 1
,
inreases.Forlowertemperaturesthehydrogenontent
ofthelmsinreasesasindiatedbytheN-Hstrething
bandat3250m 1
. Thedepositionratealsoinreased
fordereasingtemperatures,varying from 9x10 3
to
3.3 x 10 1
A/s. The maximum sample thikness
at-tainedfor6hrsdeposition was0.8m.
198 196 194 192 190 188 186 184 182
Subtrate
temperature
BN samples
F(N
2
): 44
F(B
2
H
6
): 44
B
2
H
6
/N
2
: 1x10
-2
rf-power: 0.5 W/cm
2
Intensity (u.a.)
340
0
C
180
0
C
RT
B1s
Binding Energy (eV)
Figure1. XPSspetra,showingtheBoron1sore levelfor
samplesgrownatdierentsubstratetemperatures.
500
1000
1500
2000
2500
3000
3500
4000
For all BN samples
F(N
2
): 44
F(B
2
H
6
): 44
B
2
H
6
/N
2
: 1x10
-2
rf-power: 0.5 W/cm
2
N-H
h-B-N
h-B-N
WAVENUMBER (cm
-1
)
ABSORBANCE (a.u.)
340
o
C
180
o
C
RT
o
C
dier-Theaveragegrainsize ofthehexagonalrystallites
wasalsoestimatedthroughRamanSpetrosopy. The
Raman spetra of hexagonal BN samples present an
absorptionband in1380 m 1
,aordingto thestudy
byNemanih et. al. [17℄it is possibleto estimatethe
average grain size of the nano-rystals present in the
samplesusingthefollowingrelation:
1=2 =
1447
L
a
+8:70 (2)
Where
1=2
isaFWHMoftheLorentzianurve
t-tingtheabsorptionbandandL
a
representsthe
equiva-lentgrainsizeof thenano-rystalspresentinthe
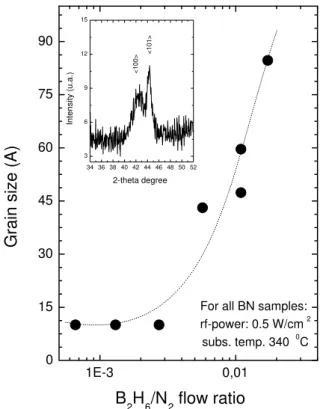
sam-ple. Fig. 3presentstheresultsobtainedapplyingthis
equationtosamplespreparedatthesameonditionbut
hangingthediboraneow. Asitanbeobserved,the
estimated averagegrain size ofthe h-BN thin lm
in-reaseswhentheB
2 H
6
owinreases. Intheinsetthe
XRD spetra for the sample with 50
A of rystalline
size is shown and peaks for 42.3and 44,3degrees,
as-soiatedtothe<100>and <101>diretionsofBN
hexagonalphaseareevident.
1E-3
0,01
0
15
30
45
60
75
90
34 36 38 40 42 44 46 48 50 52
3
6
9
12
15
For all BN samples:
rf-power: 0.5 W/cm
2
subs. temp. 340
0
C
Grain size (A)
B
2
H
6
/N
2
flow ratio
<101>
<100>
Intensity (u.a.)
2-theta degree
Figure3. Grainsize asafuntionofthesubstrate
temper-atureoftheBNsamplesobtainedusingtheequation2. In
insetaXRDspetraoftheequivalent50
Aofgrainsize.
Theresultsfor theintrinsistressfortheBN
sam-ples are shown in Fig. 4 (b), as it an be observed,
the stress presents aminimum for substrate
tempera-turearound180 0
C.ThehardnessandYoungModulus
forthe samehexagonalBNsamplesare shown in Fig.
opposite behavior ompared to the stress as the
sub-strate temperature inreases. In ontrast, the Young
modulus doesnot present an evidentorrelation with
thesubstratetemperature.
0,8
1,2
1,6
2,0
2,4
0
8
16
24
32
40
0
50
100
150
200
250
300
350
400
-0,8
-0,6
-0,4
-0,2
0,0
0,2
hardness
Young modulus
Young Modulus (GPa)
Hardness (GPa)
BN samples
F(N
2
): 44
F(B
2
H
6
): 44
B
2
H
6
/N
2
: 1x10
-2
rf-power: 0.5 W/cm
2
Intrinsic stress (GPa)
Substrate temperature (
0
C)
Figure4. (a)Hardness andYoung Modulus as funtionof
substrate temperatures for the BN samples, and (b) BN
samplesintrinsistressasfuntionofthesubstrate
temper-ature.
IV Disussion.
Infrared absorption spetrosopy is the most widely
used method for BN lms strutural haraterization
sine the hybridization states, sp 2
or sp 3
, of the
B-Nbondareeasilydistinguishedthroughwell-identied
absorption's bands. Theh-BN andturbostati phases
are haraterizedby twoinfrared ative TOphonons,
givingastrongandasymmetrialabsorptionbandE
1u
at about 1380 m 1
attributed to in-plane strething
and aweakerband A
2u
near 800m 1
resultingfrom
an out-of plane B-N-B bending mode [16℄. Thus the
resultsshowningure2ombinedwiththoseingures
1and3indiatethatitisalsopossibletogrow
Hexag-onal BN sampleswith good struture and largegrain
size bythePECVDtehniqueatlowtemperatures.
Itwasobservedthatthefundamentalparametersto
induelargegrainsizearetheB
2 N
6 /N
2
owratioand
low rf power[12, 13℄. In the literature it is observed
that the bulkvalues ofthe mehanial onstants suh
o-perpendiulartothesubstratesurfae[6℄areverylose
to theexhibited byourhexagonalBNsamples.
The values obtained for the hardness, the elasti
modulusandthethermalexpansionoeÆientare
om-parable with those reported for h-BN. The hardness,
andstresspresentoppositebehaviorwiththesubstrate
temperature,thehardnessexhibitsamaximumandthe
internalstressaminimumaround180 0
C.Atthispoint
more study is neessary to understand this behavior,
butitispossibletogetahardesth-BNthinlmatlow
temperatures.
V Conlusions
Boron nitride thin lms with dierent struture were
obtained by the PECVD tehnique. The fration of
hexagonal and amorphous phases in the thin lms is
extremelydependentontheB
2 H
6
owandH
2
dilution.
The rystallite size forthe hexagonal grains inreases
forinreasingtemperaturesandB
2 H
6
owaswellasfor
lowrf powerdensity. Themeasured thermo
mehani-al propertiesof theBNsamples didnotshowserious
variationswith thedeposition onditions. Inaddition,
thehexagonalBNnano-rystalspresentinthesamples
arewellorientedandof goodsize.
Aknowledgments
TheauthorsaregratefultoDrs. M.Tabaknis,F.L.
FreirefortheRBSand ERDAmeasurements,andDr.
M. Temperini, Dra. Nelia Ferreira, and A.C. Costa
forRamanandVisible equipmentfailities. Thiswork
was supported by Brazilian ageny FAPESP, pro #
98/01766-5.
Referenes
[1℄ C.Rohr, J.-H.Boo,andW. Ho,Thin SolidFilms, 9,
[2℄ M.Hubaek, M.Uki,T.Sato,and V.Brozek,
Ther-mohimiaAta,359,282&283(1996).
[3℄ T. Wittkowski, J. Jorzik, K. Jung, and B.
Hiller-brands,ThinSolidFilms,137,353(1999).
[4℄ J.-L. Huang, Ch-H. Pan, and D.-F. Lii, Surfae and
CoatingsTeh.166,122(1999).
[5℄ V.L.Solozhenko,D.Hausermann,M.Mezouar,andM.
Kunz,App.Phys.Lett.1691, 72(14),(1998).
[6℄ Properties of GroupIII Nitrides, editedby James H.
Edgar,KansasStateUniversity,1994,publishedby
IN-SPEC,theInstitutionofEletrialEngineers,London,
UnitedKingdom.
[7℄ Y.Kimura,T.Wakabayashi,K.Okada,T.Wada,and
H.Nishikawa,Wear,199,232(1999).
[8℄ R.Riedel,Adv.Mater.6,549(1994).
[9℄ R.T.PaineandC.K.Narula,Chem.Rev.90,73(1990).
[10℄ E.Yamaguhi,Mater.Si.Forum54-55,329(1990).
[11℄ Synthesis and Properties of Boron Nitride, edited by
J.J.Pouh and S.A Alterovitz (Trans. Teh., Zurih,
1990).
[12℄ M.N.P. Carre~no, J.P.Bottehia, and I.Pereyra, Thin
SolidFilms,308-309,219-222(1997).
[13℄ J.Vilarromero,M.N.P.Carre~no,andI.Pereyra.Thin
SolidsFilms, 373,(1-2)273-276SEP 32000.
[14℄ R.W.Homan,inPhysis onthinlms,editedbyG.
Hass and R.E. Thun (Aademi, New York, 1966),
vol3.
[15℄ J. F. Moulder, W. F. Stikle, P. E. Sobol, and K.
D. Bombem, Handbook of X-ray Photoeletron
Spe-trosopy, edited by J. Chastain, Perkin Elmer Co.,
1992.
[16℄ D.M.Homan,G.L.Doll,andP.C.Eklund,Phys.Rev.
B6051,30(10),(1984).
[17℄ R.J. Nemanih, S.A. Solin, and R.M. Martin, Phys.