r e v b r a s r e u m a t o l . 2017;57(5):487–490
w w w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Case
report
Improvement
of
nailfold
capillary
microangiopathy
after
immunosuppressant
therapy
in
a
child
with
clinically
amyopathic
juvenile
dermatomyositis
Melhoria
na
microangiopatia
capilar
periungueal
após
terapia
imunossupressora
em
uma
crianc¸a
com
dermatomiosite
juvenil
clinicamente
amiopática
Lúcia
Maria
Arruda
Campos
a,∗,
Adriana
M.E.
Sallum
a,
Cintia
Z.
Camargo
b,
Luís
Eduardo
C.
Andrade
b,
Cristiane
Kayser
baUniversidadedeSãoPaulo(USP),FaculdadedeMedicina,UnidadedeReumatologiaPediátrica,HospitaldasClínicas,Institutoda
Crianc¸a,SãoPaulo,SP,Brazil
bUniversidadeFederaldeSãoPaulo(UNIFESP),DepartamentodeReumatologia,SãoPaulo,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received30November2014 Accepted15March2016 Availableonline4June2016
Introduction
Juveniledermatomyositis(JDM)isararediseasethatbelongs tothegroupofidiopathicinflammatorymyopathies.1
Clin-icallyamyopathicdermatomyositis(CADM)isanevenrarer entity in pediatrics, with only 75 cases described in the literature.2CADMpatientspresentmildornomuscle
involve-mentandthecutaneousmanifestationsareindistinguishable fromthoseseeninclassicaldermatomyositis(DM).3,4
Systemicinflammatoryvasculopathyisanimportant char-acteristicof JDMaffecting especiallythe microcirculation.5
Nailfold capillaroscopy (NFC) is a non-invasive method that allows the direct visualization of nailfold capillaries.6
Decreasednumberofcapillaryloops(devascularization) asso-ciatedwithenlargedcapillariesandbranchingcapillaryloops
∗ Correspondingauthor.
E-mail:[email protected](L.M.Campos).
arethemostcharacteristicfindingsobservedinJDM.7In
addi-tion,severalstudieshavedescribed anassociationbetween NFC abnormalities and JDM severity and activity.8 To the
bestofourknowledge,NFCabnormalitieshavenotbeen sys-tematically studied inCADM. We describeherein the case of a 4-year-old child diagnosed with juvenile CADM with importantchangesinNFC,whoseresponsetotreatmentwas followedbysignificantimprovementincapillaroscopy abnor-malities.
Case
report
InJune2008,a4-year-oldgirlwasattendedwithafourmonths complaintofmalarrash,photosensitivity,anderythematous lesionsovertheproximalinterphalangealjoints,elbowsand
http://dx.doi.org/10.1016/j.rbre.2016.05.002
488
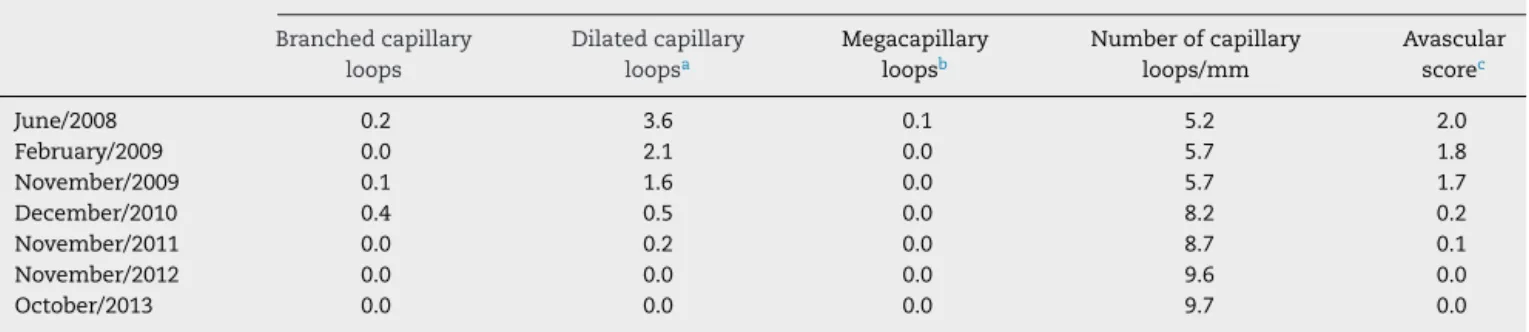
rev bras reumatol.2017;57(5):487–490Table1–SequentialcapillaroscopicevaluationperformedfromJune2008toOctober2013inapatientwithJuvenile AmyopathicDermatomyositis.
Capillaroscopyparameters(meanvaluefortenfingers)
Branchedcapillary loops
Dilatedcapillary loopsa
Megacapillary loopsb
Numberofcapillary loops/mm
Avascular scorec
June/2008 0.2 3.6 0.1 5.2 2.0
February/2009 0.0 2.1 0.0 5.7 1.8
November/2009 0.1 1.6 0.0 5.7 1.7
December/2010 0.4 0.5 0.0 8.2 0.2
November/2011 0.0 0.2 0.0 8.7 0.1
November/2012 0.0 0.0 0.0 9.6 0.0
October/2013 0.0 0.0 0.0 9.7 0.0
a Thethreelimbsofthecapillaryloopshouldbeenlargedover4times.
b Thethreelimbsofthecapillaryloopshouldbeenlargedover10times.
c Avascularareaisdefinedasthelackofmorethantwosuccessivecapillaryloops;grade0(noavasculararea);grade1(oneortwodiscrete
avascularareas);grade2(morethantwodiscreteavascularareas);grade3(extensiveandcoalescentavascularareas).
knees,withnocomplaintsregardingmusclestrengthlossor pain. The ManualMuscle Testing(MMT)9 score was 80/80,
Childhood Myositis Assessment Scale (CMAS)9 was 48/52,
muscularDiseaseActivityScore(DAS)was2/11andcutaneous DASwas6/9.10Laboratorytestsshowedhemoglobin13.6g/L,
hematocrit38.5%,leukocytes21,000/mm3 (76%neutrophils,
16%lymphocytes), platelets289,000/mm3,erythrocyte
sedi-mentationrate(ESR)23mm/1sthour,C-reactiveprotein(CRP) undetectable,aldolase20.8IU/L(normal<7.6),creatinekinase (CK) 130IU/L (normal<204), lactate dehydrogenase (LDH) 575IU/L (normal<480), aspartate aminotransferase (AST) 29IU/L(normal<34),alanineaminotransferase(ALT)14IU/L (normal<44),andpositiveantinuclearantibody(1/640, homo-geneousfinespeckledpattern).Capillaroscopywasperformed inallfingersofbothhandsusingastereomicroscope (Olym-pusSZ40)at10×to16×magnificationunderepi-illumination at45◦,analyzingnumberofcapillary/mm,enlarged,giantand
branched capillaries, and avascular score.11 Capillaroscopy
showed a scleroderma (SD) pattern, with severe microan-giopathy,characterizedbydecreasednumberofthecapillary loops with intense avascular areas, few branching and frequentdilatedcapillaryloops(Table1;Fig.1).Clinically amy-opathicdermatomyositis(CADM)diagnosiswasestablished,3
since Bohan and Peter criteria were not fulfilled.12
Treat-mentwithprednisolone(0.5mg/kg/day),hydroxychloroquine (5mg/kg/day),andphotoprotectionwasthenintroduced.
Afterfourmonths,thepatientpersistedwithdisease activ-ity. Muscle magneticresonance imaging(MRI) and muscle biopsywere normal.Electromyography wasnotperformed. Hydroxychloroquine dose was increased, and methotrex-ate(0.5mg/kg/week),folicacidand topicaltacrolimuswere associated, withpartial improvement ofsymptoms, which allowedgradualreductionofprednisolonedose.InJuly2009, the patient started complaining of fatigue and new Got-tron’spapules andelevationofLDH serumlevels (666IU/L) were observed. Prednisolone and methotrexatedoses were increasedandthalidomide(50mg/day)wasintroducedwith satisfactoryresponse.InOctober2009,thepatientwas con-sideredinclinical remission,withMMT80/80,CMAS48/52, muscularDAS1/11,cutaneousDAS0/9,normalESRandCRP, aldolase8.7IU/L,CK64IU/L,LDH524IU/L,AST22IU/L,andALT 18IU/L.Capillaroscopypresentedslightimprovement.Atthis
moment,hydroxychloroquinewasdiscontinued,followedby withdrawaloftopicaltacrolimus(February2010),prednisolone (April2010),methotrexate(December2010),andthalidomide (July2011).
At her last appointment (October 2013), the patient remained on remission, MMT 80/80, CMAS 51/52, muscu-lar DAS 0/11, cutaneous DAS 0/9, normal ESR and CRP, aldolase5.1IU/L,CK115IU/L,LDH400IU/L,AST20IU/L,and
rev bras reumatol.2017;57(5):487–490
489
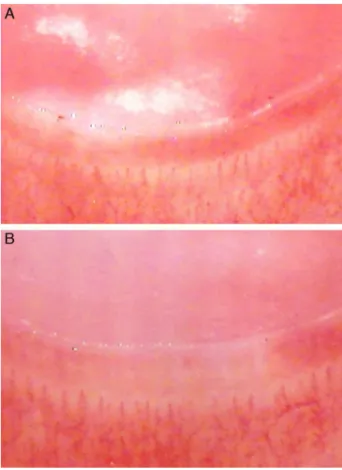
Fig.2–Capillaroscopy(October,2013).Nailfold
capillaroscopyshowinganormalpattern,withanormal numberofcapillaries/mm,nodilatedormegacapillaries andrevascularizationofavascularareasinthesecondand fourthdigitfromthelefthandrespectively(15×
magnification)(Fig.2AandB).
ALT14IU/L.Capillaroscopywasnormal,showingsignificant improvementinthemicroangiopathy,normalnumberof cap-illaries/mm,noavascularareasandnobranchingordilated capillaryloops(Table1;Fig.2).
Discussion
Thisisthefirstreportonthepresenceofexuberant periph-eralmicroangiopathyevaluatedbyNFCinachildwithCADM, followedbyaprogressiveandsignificantimprovementofNFC changesaftersuccessfulimmunosuppressivetreatment.
Although there are not studies reporting the dynamic natureofNFCmicroangiopathicabnormalities (dilated cap-illary loops, megacapillaries and avascular areas) and its correlation with disease activity in CADM patients, as describedinthecasepresentedherein,thisaspecthasbeen observedinseveralstudiesincludingclassicalJDM.7,8
Capil-laroscopywasevaluatedin61JDMchildrenover36months. Theimprovementinthenumberofloops/mmwasassociated withlessintensecutaneousactivityandmonocyclicdisease course.Ontheotherhand,polycyclicdiseasewasassociated withmaintenanceofcutaneousactivityandpersistent cap-illaroscopychanges.TherewasnocorrelationbetweenNFC andmusclediseaseactivity,suggestingthattherearedifferent
mechanismsunderlyingthepathogenesisofskinandmuscle inJDMvasculopathy.13AlthoughNFCabnormalitiesarenot
includedinJDMclassificationcriteria,12theyseemtoreflect
systemicvasculopathy,andsomeauthorssuggestits inclu-sionamongclassificationcriteriaforJDM.14SinceCADMcould
beconsideredpartofthespectrumofJDM,itcouldbeassumed thatbothsituationssharethesameNFCbehavior.
Inthepresentcase,thepatientwasdiagnosedasCADM, most specifically the subphenotype denominated juvenile hypomyopathicdermatomyositis (HDM), which designation is appliedfor patients with cutaneousDM and no clinical evidence of muscle disease (i.e. weakness), whose sub-clinical evidence of myositis upon laboratory (e.g. muscle enzymes),electrophysiological,and/orradiologicalevaluation isfoundduringinvestigation.3,4Infact,shepresentedmajor
skindiseasemanifestationsassociatedwithslightincreased muscle enzymes (aldolase and LDH), but without muscle complaintsorweakness.Muscularactivityscoreswere con-sidered aswithinthenormalranges.15 Thesmalldecrease
in CMAS observed in this patient at disease presentation may beattributedtoherearlyage andlackofcooperation in carrying out some of the exercises required. The nor-mal results of MRI and muscle biopsy could be partially affected bythelow-dosecorticosteroiduse, sincethis ther-apyhadbeeninitiatedfourmonthsbeforethesetestswere performed.
Inareviewdescribing68casesofjuvenileCADM,56%was classifiedasamyopathicDM(ADM),18%asHDMand26% pro-gressedtoclassicalJDMafteramedianfollowupof1.9years. However,itwasnotpossibletodeterminetheparametersthat couldpredicttheoutcomeofpatientswithCADMtothe clas-sic formofJDM. Calcinosiswaspresent inless than5%of thecasesandnochildrenhadcutaneousorgastro-intestinal vasculitis,pulmonaryinfarction,interstitialpneumonitisor malignancy,whichsuggestsagoodprognosisforthisvariant ofJDM.2
The best therapeutic option inCADM is still controver-sial, since there are no randomized studies in this field. Some authors argue that early use of corticosteroids and immunosuppressivedrugscouldpreventprogressiontoJDM. Alternatively, in areview ofjuvenile CADM treatment, the authors concluded thatonly photoprotection,topical med-ications and oral hydroxychloroquine should be initially used and corticosteroids and immunosuppressant therapy shouldbereservedforrefractorycases.2,16 Ourpatient
pre-sentedsevereskinmanifestationsandtheuseofconcomitant photoprotection, topical therapy, hydroxychloroquine and immunosuppressant drugswasnecessary.However, resolu-tionoftheskinandcapillaroscopicabnormalitieswasmore clearlyattainedaftertheintroductionofthalidomide.Indeed, theefficacyofthalidomideinthetreatmentofrefractoryforms ofJDM,aswellinCADM,hasbeenpreviouslydescribed.17
490
rev bras reumatol.2017;57(5):487–490Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1. SatoJO,SallumAM,FerrianiVP,MariniR,SacchettiSB,Okuda EM,etal.,RheumatologyCommitteeoftheSãoPaulo PaediatricsSociety.ABrazilianregistryofjuvenile dermatomyositis:onsetfeaturesandclassificationof189 cases.ClinExpRheumatol.2009;27:1031–8.
2. GeramiP,WallingHW,LewisJ,DoughtyL,SontheimerRD.A systematicreviewofjuvenile-onsetclinicallyamyopathic dermatomyositis.BrJDermatol.2007;157:637–44.
3. SontheimerRD.Wouldanewnamehastentheacceptanceof amyopathicdermatomyositis(dermatomyositissiné myositis)asadistinctivesubsetwithintheidiopathic inflammatorydermatomyopathiesspectrumofclinical illness?JAmAcadDermatol.2002;46:626–36.
4. GhaziE,SontheimerRD,WerthVP.Theimportanceof includingamyopathicdermatomyositisintheidiopathic inflammatorymyositisspectrum.ClinExpRheumatol. 2013;31:128–34.
5. SallumAM,MarieSK,WakamatsuA,SachettiS,ViannaMA, SilvaCA,etal.Immunohistochemicalanalysisofadhesion moleculeexpressiononmusclebiopsyspecimensfrom patientswithjuveniledermatomyositis.JRheumatol. 2004;31:801–7.
6. SatoLT,KayserC,AndradeLE.Nailfoldcapillaroscopy abnormalitiescorrelatewithcutaneousandvisceral involvementinsystemicsclerosispatients.ActaReumatol Port.2009;34:219–27.
7. NascifAK,TerreriMT,LenCA,AndradeLE,HilárioMO. Inflammatorymyopathiesinchildhood:correlationbetween nailfoldcapillaroscopyfindingsandclinicalandlaboratory data.JPediatr.2006;82:40–5.
8. OstrowskiRA,SullivanCL,SeshadriR,MorganGA,Pachman LM.Associationofnormalnailfoldendrowloopnumbers withashorterdurationofuntreateddiseaseinchildrenwith juveniledermatomyositis.ArthritisRheumatol.
2010;62:1533–8.
9.LovellDJ,LindsleyCB,RennebohmRM,BallingerSH,Bowyer SL,GianniniEH,etal.,TheJuvenileDermatomyositisDisease ActivityCollaborativeStudyGroup.Developmentofvalidated diseaseactivityanddamageindicesforthejuvenile
idiopathicinflammatorymyopathies.II.TheChildhood MyositisAssessmentScale(CMAS):aquantitativetoolforthe evaluationofmusclefunction.ArthritisRheumatol.
1999;42:2213–9.
10.BodeRK,Klein-GitelmanMS,MillerML,LechmanTS, PachmanLM.Diseaseactivityscoreforchildrenwithjuvenile dermatomyositis:reliabilityandvalidityevidence.Arthritis Rheumatol.2003;49:7–15.
11.AndradeLEC,GabrielAJr,AssadRL,FerrariJAL,AtraE. Panoramicnailfoldcapillaroscopy:anewreadingmethodand normalrange.SeminArthritisRheum.1990;20:21–31.
12.BohanA,PeterJB,Polymyositisdermatomyositis.NEnglJ Med.1975;292:344–7.
13.Christen-ZaechS,SeshadriR,SundbergJ,PallerAS,Pachman LM.Persistentassociationofnailfoldcapillaroscopychanges andskininvolvementoverthirty-sixmonthswithdurationof untreateddiseaseinpatientswithjuveniledermatomyositis. ArthritisRheumatol.2008;58:571–6.
14.BrownVE,PilkingtonCA,FeldmanBM,DavidsonJE,Network forJuvenileDermatomyositis,PaediatricRheumatology EuropeanSociety(PReS).Aninternationalconsensussurvey ofthediagnosticcriteriaforjuveniledermatomyositis(JDM). Rheumatology(Oxford).2006;45:990–3.
15.HasijaR,PistorioA,RavelliA,DemirkayaE,KhubchandaniR, GuseinovaD,etal.,PediatricRheumatologyInternational TrialsOrganization.Therapeuticapproachesinthetreatment ofjuveniledermatomyositisinpatientswithrecent-onset diseaseandinthoseexperiencingdiseaseflare:an
internationalmulticenterPRINTOstudy.ArthritisRheumatol. 2011;63:3142–52.
16.WallingHW,GeramiP,SontheimerRD.Juvenile-onset clinicallyamyopathicdermatomyositis:anoverviewofrecent progressindiagnosisandmanagement.PaediatrDrugs. 2010;12:23–34.