Changes in Aggregate Form, Size and Flexibility Along
Phase Sequenes in Lyotropi Liquid Crystals
Lia Queirozdo Amaral
InstitutodeFsiadaUniversidadedeS~aoPaulo,
C.P.66318,S~aoPaulo,SP,05315-970, Brazil
Reeivedon14November,2001
Aomparisonismadebetweenexistingtheoriesforself-assemblingsystemsandexperimentalphase
diagrams of omplexsystems made up of amphiphile/water/additives, with emphasis on phase
transitions by hangesinonentration. Evidenefor dierent typesofhexagonal (H) phases,in
systemswithdetergentandlipidamphiphiles,arereviewed. ItisshownthatharateristisoftheH
phaseandofthephasesequene(ubiphaseswithlipidsandnematiphaseswithdetergents)are
revealedthroughtheexponentdeningthevariationofthehexagonalparameterwithamphiphile
onentration. Emphasisis also givento results obtained in the ternary system sodium dodeyl
(lauryl) sulfate / water /deanol, whihexhibits the phase sequeneisotropi (I) -H -nemati
ylindrial(N){[nematibiaxial (Nb)℄-nematidisoti (Nd)-lamellar(L), witho-surfatant
addition. Existingtheoriesfor self-assemblingsystemsofrigidandexiblerodspredit thephase
sequeneI -(N
) -H as afuntion of inreased partile volume fration
p
, with atriple point
separatingI-HandI-N -Hphasesequenes. Possiblereasonsforthe non-trivialexperimental
I-H -N
inversioninphasesequeneare disussed. A omplexpaththroughthe phasediagram
is able to explain the experimental results interms of hanges inmiellar growth and exibility
induedbytheo-surfatant. Theroleofthesurfatantparameter,thatexpressestheurvatureof
thepolar-apolarinterfaes,explainsmuhoftheobservedbehavior,inludingtheN{Ndtransition
withinreaseindeanol/amphiphilemolarratio.
I Introdution
The omplex polymorsm of systems made of
am-phiphile / water / additives (salt and / or alohol)
hasbeenextensivelyinvestigatedoverthelastdeades
[1,2℄. Suh polymorsm existsbeausetheamphiphile
moleules in solution self-assembly into aggregates of
dierentformsandsymmetries,duetothetendenyof
spontaneousformationofinterfaesbetweenpolarand
apolargroups. Theformationoftheaggregatesis
on-trolledbythesurfatantparameter[3℄p
o
=v=al,with
v,aandl,being,respetively,thevolumeofthehain,
the polar head area at the interfae (whih depends
also on theamount of bound water) and theeetive
lengthofthehain. Moleuleswith p
o
=1/3,1/2and
1havetendenyto form,respetively,spherial,
ylin-drialandplanarmielles. Inamphiphileswithasingle
hydroarbonhain (as detergents,with p
o
1/3) the
interfaes may be losed and highly urved, with
for-mationofmielles (diretin thewaterrih sideofthe
phasediagram,invertedinthewaterpoorside)and
sep-arationofhydroarbonandwatermoieties. Miellesof
initially spheroidalshape (whenformed at theritial
miellaronentration) may growto ylindrial forms
quent derease in p
o
due to derease of bound water.
Lamellarphases with planarinterfaes oromplex
bi-ontinuous phases, with minimal urvature interfaes,
may also our in the intermediate zone of the phase
diagram. Amphiphiles with two hydroarbon hains
(as phospholipids, with p
o
1), instead, have
ten-deny offorming vesiles(losed membraneswith
wa-terinsideandoutside)duetotheirdiÆultyinforming
highly urved interfaes. Suh supra-moleular
aggre-gates mayhaveornot longdistane order,givingrise
toisotropiandliquid-rystallinephases,before
reah-ing rystalline phases. Changes in the miellarshape
and in the separatinginterfaes aompany or trigger
thephasetransitionsasafuntionofonentrationand
temperature.
We shall here onentrate on amphiphiles with a
single arbon hain, and onphase transitions indued
by hanges in onentration. The typial phase
se-quene with inreasing amphiphile onentration is I
(isotropi) - H (hexagonal) - L (lamellar).
Interme-diate phases between H and L, with long-range
posi-tional order, may our in binary systems.
I-Q-H sequenes)or of biontinuous arrays(in H-Q-L
sequenes). Ternary systems, with addition of a long
hainalohol,mayleadinsteadtonemati(N)domains
betweenHandLphases(ordiretlyfromIphase),with
uniaxial[5-7℄(ylindrialN
anddisotiN
d
)and
biax-ial[8℄(N
b
)phaseshavingonlylong-rangeorientational
order. Therole ofaloholin hangingtheaggregation
proess,leadingtoislandsofnematiphasesinternary
phase diagrams,ishere disussed. The harateristis
ofHphasesseemalsotodependonthepartiularphase
sequene where it appears. In this paper we shall
re-view thework we havemade on suh phases overthe
last deade,withseveralollaborators.
SinethedisoveryofHphases,theirstruturehas
beenproposed[1℄toonsistof\innite"ylindrial
mi-elles with a two-dimensional positional order in the
planeperpendiularto theylinderaxes,basedonthe
fatthatonly(hk0)dirationswereobserved,without
dened values for the parameter, together with the
indiationofverylongmiellesbyothertehniques,as
NMR. Duringthelastdeade, however,wehave
gath-ered evideneshowingthat in fat dierent strutures
exist, whih seem to orrelate with the spei phase
sequene. Information on suh strutures is given by
theexponentofthevariationof thehexagonal
param-eter with onentration. Results obtained [9-13℄ with
detergentandlipidamphiphiles areherereviewed.
Wefousalsointhispaperontheanalysisofresults
obtained[14-17℄inthesystemsodiumdodeyl(lauryl)
sulfate(SLS)/water/deanol(whihisagood
exam-ple [18-20℄ofdetergentbehavior). Thephasesequene
I-H-N
{(N
b )-N
d
-L,obtainedwithinreaseinthe
deanol /amphiphilemolarratio (M
d
),is analyzedin
partiular. The resultsare onfrontedwith statistial
mehanialtheories[21-23℄ofself-assemblythatpredit
insteadthephasesequenesI-N
-HorI-N
d
-L,
de-pending onthe symmetryof theaggregates(ylinders
ordiss,respetively). Weonsideralsoareentresult
[24℄obtainedthroughanapproahbasedonbending
en-ergyonsiderations,whih isabletoaountforshape
transformationsattheN
-N
d
transition,withinrease
ofdeanolontentinsystemswith mixedmielles.
II Oupany of the hexagonal
ell
Asanalyzedinourpreviouspapers[9,11-13℄,fora
sphe-roylinder (SC) of radius R and total length L in an
hexagonalellwithparametersaand,theusual
on-dition of equal densityat the mirosopi and
maro-sopilevelsimpliesthatthestatistruture(averaged
in time)satisestherelation
(L=)
SC =(2
p
3=)
v (a=2R )
2
()=( 1=3); (1)
one for spheres) and
v
is the volume fration of the
totalamphiphile moiety. Note that in the aseof
mi-ellestheexisteneofboundwatermaymake
v
dier-entthan thepartile volume fration
p
. For longSC
theylinder (or rod) limit is obtained,and the fator
/(-1/3),thatdependsontheanisometry,beomes
1. ThislimitanbeusedinordertoobtainL=values
in the H phase, if mielles have largebut unknown
values[13℄.
Furthermore,intheaseofmielles,asstudiedhere,
aremustbetakeninthedenitionoftheRandL
val-ues in Eq. (1). They may refer to \eetive" radius
R
eff
andlengthL
eff
,ofthe\partilewithoutwater",
relatedto
v
. Tosubstitute
p for
v
inEq. (1)itwould
beneessaryto useR
tot andL
tot
ofthe\partile",
in-luding bound water. Alternatively, one an onsider
theparaÆnimielleandthevaluesin Eq. (1)beome
R
par , L
par and
vpar
, where
vpar
is referredtoas the
paraÆnivolume fration. Moreoverthefollowing
re-lationshipisvalid[13℄:
(R
e =R
par )
3
=(
v =
vpar )(
par
1=3)=(
e
1=3) (2)
ForlongmiellesthedierenesbetweenL
e ,L
par ,
L
tot
maybenegleted,but notthedierenesbetween
therespetiveRvalues.
IntheseHphasesnounique valueexists,beause
ofpolydispersityof miellarlength, aneessary
ondi-tion for the ourrene of diret I-H phase transition
[21℄,but \averagevalues" aremeaningful. Asweshall
see,thebehavioroftheHphaseandofphasesequenes
dependsontherelationsbetweenaand. Inaseof
-nitemiellesanduniformdereasein inter-partile
dis-tanes in all three dimensions, the anisometry of the
partileexistsalsointheelland oneexpets:
L==2R =a (3a)
Inaseof\innite"miellesoneexpetsinstead:
L==1 (3b)
The struture of the H phase, and partiularly its
evolutionwithinreasedonentration,willdependon
whih Eq. (3) is valid for the partiular system, and
onthefat thatLmaybeaonstantwithintherange
oftheHphaseormayvary withonentration. When
Eq. (3a) applies and the L value is onstant, it
re-sults a 1=3
v
. When Eq. (3b) applies, it results
a 1=2
v
. In the general ase, if it is experimentally
observed a =A x
v
, theondition given by Eq. (3a)
leads[11℄toanequationthatexpressesthevariationof
theanisometrywithonentration:
=[3 3K (1 3x)
v ℄
1
; (4)
p
Forylinderswithonstantradius,Kisaonstant.
Forx=1/3,theanisometryisalsoaonstant,givenby
Eq. (4). Butifx <1=3,Eq. (4) givestheformof
mi-ellar growthwith onentration, under the ondition
of Eq. (3a). The funtion K (1 3x)
v
varies from 2/3
to1as variesfrom 1(sphere)to1. However,asthe
limitofinniteylinderapproahes,Eq. (3a),andalso
Eq. (4),arenolongervalid,andtheonditiongivenby
Eq. (3b)isapproahed.
Thevalidityof Eq. (3)forapartiular systeman
beanalyzed[11℄throughomparisonofvaluesL=,
ob-tainedfrom Eq. (1)under thehypothesis oflongrods
(anisometryfator=1),and 2R =a,usinganestimated
Rvalueandtheknownaand
v
values. IfL=<2R =a;
there is indiationof shortrods, when theanisometry
fatorofEq. (1)(always<1)annotbenegleted. But
L=>2R =aindiates abreakdownofEq. (3a).
It has been also theoretially shown, in statistial
mehanis treatment of self-assembly systems [21,22℄,
that theexponents1/3and 1/2hold, respetively,for
niterigidrodsandforlongexiblerods(with
end-to-end ontat orwith persistent length on the order of
). Weshall thereforereferto onditionsL/ =2R/a
andL==1,Eq. (3),asharateristiof\nite/rigid"
and\innite/exible"rods,respetively. Intheaseof
exible objets Eq. (1) does not hold in strit sense,
but it is always possible to obtain an \apparent L=
value"whihsatisesEq. (1)forrods.
III The H phase in dierent
sys-tems
InthebinarySLS/watersystemtheintervalofthe
ex-plored
v
values,withintheHdomain, wassuÆiently
largeandallowedus tounambiguouslydene[9℄a1/3
behavior,typialof niterigidrodswithonstant
ani-sometry. Apreviousstudy[25℄oftheI-Hphase
transi-tionhadshownthattheylindergrowthdoesnotour
within the I phase,but at thetransition itself, aview
onsistent with the statistial theories published later
[21℄. Adetailedstruturalstudy[13℄withareful
anal-ysisoftheeletrondensitymapsobtainedfrom
dira-tionintensitiesonrmednite, althoughlong,rodsin
theHphaseofthebinarySLS/watersystem.
Further studies in other amphiphile systems
evi-dened that the exponent depends on the partiular
phasesequene,being1/2inasystemwitha
neighbor-ingholesteriphase[10℄ andsmallerthan1/3whena
neighboringubiphaseispresentinlipids[11,12℄. For
OLPC (oleoyl-lyso-phosphatidil-holine) / water
sys-tem,withI{H{Q
b
(ubibiontinuous){Lphase
se-quene,thegrowthofthelipidmielles,obtainedfrom
Eq. (4),waslearly followedalong theHdomain [11℄,
withanisometrynear1attheI-Htransitionupto1
at the H { Q
b
transition. The strutures of H and Q
phosphatidyl-holine),withI-Q
m
(ubimiellar){H
-Lphasesequene,andDTAC(dodeyl-trimethyl
am-monium hloride), with I { Q
m
{ H { Q
b
{ L phase
sequene have been detailed analyzed [12℄. Miellar
growthwasveriedalong the H phasesof these three
lipids(exponentx<1/3)andepitaxialrelationshipsat
both Q
m
{ H and H { Q
b
phasetransitions ould be
demonstrated[12℄. MiellesaresmallafterI {H
tran-sition,largerafterQ
m
{Htransition, \innite"before
H -Ltransition andhaveevenL=>1before H{Q
b
transition.
AstudyoftheI{Q
m
transition, thatpreedesthe
Q
m
-H transition in PaLPCrevealed [26,27℄that
mi-ellesremainspheroidalanddonotgrowtoSCintheI
phase,duetothelargersurfatantparameter,andalso
howthemiellarshort-rangeorderoftheIphase
trans-formsintothemiellarubiorderduetolosepaking
of the spheroidal mielles. For SLS mielles, a
trans-formation of spheroidal into SC polydisperse mielles
ours [28℄ stillin theI phase, dueto thesmaller
sur-fatantparameter,preparingtheI-Hphasetransition.
Therole ofthesurfatantparameterin promoting
ag-gregatetransformationswill befurther disussedlater
on.
Thepiturethatemergesfromthesestudiesfollows
a pattern. In the harged detergent SLS, miellesare
already small SC in the I phase, and grow at the I{
H transition, due to the\orientationindued growth"
mehanism [21℄, but have afterwards a onstant size,
untildistortionofthemiellarunitinduesphase
tran-sitionsleadingnallytotheLphase. Inneutrallipids,
growthdoesnotouratthetransitiontotheHphase,
but moreslowlyin thewhole Hrange. Inaseswhere
an \exess growth" ours, revealed by the ondition
L= > 1 when Eq. (1) is used (probably related to
undulationsofthemiellesurfae),thetransitionis
to-wardsabiontinuous ubiphase [11,12℄,while aH-L
transition is observed when the limit L= = 1 is not
over-passed. Itisnotyet possibletoonludewhether
these dierenesbetweenharged detergentsand
neu-tral lipids are due diretly to the soft inter-miellar
Coulombinterationortodierenesinwaterbinding.
Theexisteneofanemati (orholesteri)domain
seemstoorrelatewithaneighborHphasewith
expo-nentx= 1
=
2
(innite/exibleobjets), what is
intrigu-ing,sinethelyotropinemati phasesareexpetedto
orrespondtosmall mielles[7℄,andaminimum
rigid-ity is also neessary forthe N phases [23℄. Thus, the
roleofdeanolinpromotingtheappearaneofNphases
deservespartiular attention.
IV The H phase neighbour to N
phase
sys-entrateintheN domainof theternarysystemSLS/
water /deanol [18-20℄. Thephase sequene depends
on onentration and temperature and may be I - N
or I -H- N
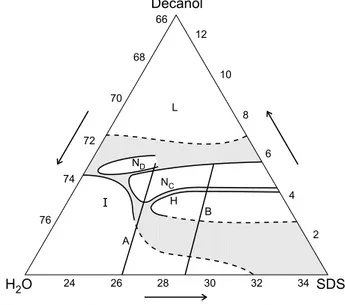
[14-16℄. Figure1showstheexperimental
phasediagram.
The
v
intervalwhen theHphase isrossedin the
I-H-N
sequene (linesAandBin Fig. 1) istoosmall
toallowthedeterminationoftheexponentx,obtained
in thebinarysystem. However,aharaterizationan
be made by adiret omparison of the L= value
ob-tainedfromEq. (1),withknown
v andR
eff =18.4
A
[9,13℄, and the 2R
tot
=a value (a=2/ p
3 s 1
), as
per-formed in refs. [15,16℄. R
tot
is the \partile" radius,
givenbythesumoftheextendeddodeylhain,16.7
A,
andthepolarheaddiameter,4.6
A.ThetwovaluesL=
and 2R
tot
/a give the best denition of the
\ylindri-al partile" ofthe H phasein the diretions anda,
respetively.
Decanol
SDS
H O
2
66
12
10
8
68
70
72
74
76
24
26
28
30
32
34
2
4
6
L
N
C
N
D
H
B
A
Figure 1. Ternary phase diagram of the system
SLS/water/deanol as a funtion of weight omposition
from Quist et al[20℄, whih inludes the nemati domain
previouslyloalizedbyAmaraletal[18,19℄ andthe limits
ofthehexagonalphasegivenbyEkwall[2℄,whentheN
do-mainwasnotknown. ResultsforlinesA[15℄andB[16℄are
presentedinFig. 2. ThenotationSDS isused for SLSin
ref.[2℄and[20℄.
Figure2(given alsoin [16℄)givesL= and2R
tot =a
values from three dierent data sets [15,16,25℄, as a
funtion of the miellar
v
, whih inludes both SLS
and deanol. The threedatasets orrespondto
dier-ent SLS:water molarratios (M
w
), two in ternary
sys-tems, withM
w
=39.4(line AofFig. 1[15℄) and45.2
(line Bof Fig. 1[16℄), and one in the binary system
(M
w
= 24.0[25℄). The rsttwopoints forM
w =39.4
orrespondto theHphaseinoexistenewithIphase.
It isevidentfrom Fig. 2that theonditionL= =
2R
tot
=a is only attained in the H phase of the
stud-ied binary SLS/water system [25℄, in agreement with
a
1=3
behavior [9℄. However L= values are lose
to1and muhlargerthan 2R
tot
=a in theH phasesof
theternarySLS/water/deanolsystem, fortwo
dier-ent valuesof M
w
[15,16℄. It is seen that the addition
ofdeanol deeply hanges the harateristisof the H
phase,whihswithesfrompartilenite/rigidto
parti-leinnite/exiblebehavior. TheresultsforM
w =39.4
showevenatendenyforinreaseinL=andsmall
de-reasein2R
tot
=awithinreaseindeanolontent.
Figure2. (Appearingalsoin[16℄): L=(solid symbol)and
2Rtot=a(opensymbols)valuesintheHphasesasafuntion
of(SLS+deanol)volumeonentrationforthreedatasets,
orresponding to three dierent values of the water: SLS
molar ratio. Squares orrespond to the results [15℄ with
M
w
=39.4,irlestoaternarysystemwithM
w
=45.2[16℄
andtriangles to abinarysystem with Mw=24.0 [25℄. L=
valueswereobtainedfromEq.(1),using
v andR
e =18.4
A.
Rtot and a are, respetively, the total partile radius (see
textfor details)andthehexagonalellparameter.
Itshould benotedthattheR
eff
valueusedto
al-ulateL=wasobtainedinthebinarysystem[9,13℄. In
orderto further demonstrate theresultin theternary
system, analysis of the eletron density maps in the
ternarysystemwerealso performed[16℄. Themaps of
theternarysystem[16℄lookedverysimilartothemaps
[13℄ofthebinarysystemregardingthemiellarobjet.
Onlythefrationoupiedbythemielleinthe
hexag-onalellhanges,aordingtodierent
v
andavalues
ofthephases. Theternarysystemhasahexagonalell
withonsiderablymorewaterin thehexagonalplane.
Aonsistentinterpretationof theylindrial
stru-tureisobtained[13℄byestimatingR
eff
fromthe
ele-trondensitymaps using asariterion thelargest
dis-taneoftheontinuousontourfrom themiellar
en-ter. Results show [16℄ that, in fat, R
eff
is slightly
smaller in the ternary system. The average value is
about 4% smaller than the value obtained in the
bi-narysystem. TheR
par
values howeverare within 1%
thesameasin thebinary system. Thisindiates that
thepreseneofdeanolhangesonlyslightlythe
miel-larradius, and that the hange in R
eff
ours in the
Asfar astheL=valuesareonerned, theyareequal
to1,withintheevaluatedunertainties. Itmeansthat
suh ternary H phasesare builtup of innite/exible
ylindersinhexagonalarray.
Inorder to larify the role of deanol, we analyze
available theoriesofself-assemblyandonfront
experi-mentalandtheoretialphasediagrams.
V Phase diagrams and theories
of self-assembly
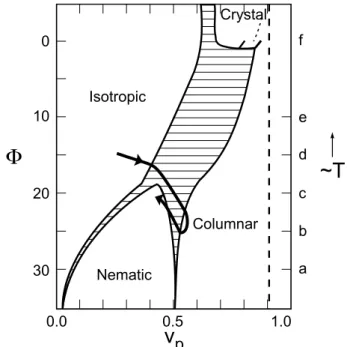
The theories of self-assembly predit the sequene I
-(N
) - H, as a funtion of inreased partile volume
fration
p
, for ases of nite hard rods [21℄,
persis-tent exible rods [22℄ and wormlikemielles [23℄. All
these theoriespresent phasediagramsasafuntion of
p
with thesametopology,seenin Fig. 3. There is a
triple point that separates I-Hand I-N-H phase
tran-sitions. In the ase of nite rigid objets, the phase
diagram[21℄isafuntionoftheassoiationfreeenergy
,whihdenesthepartilelength. Intheaseof
ex-ible longrods, thephasediagram [22,23℄is afuntion
oftheratiopersistentlength/miellediameter(P/D)
withtheaxial ratio(L/D)asparameter(D=2R).The
diret I-Htransition isexpeted in the limitsof small
rigidobjetsorveryexiblelongobjetsinIphase. In
theformeraninreaseinmiellarlengthisexpetedat
I-H transition[21℄.
TheexpetedtheoretialphasesequeneI-N
-H
ap-pearsintheexperimentalphasediagramoftheternary
system SLS/water/deanol, shown in Fig. 1, only for
about4wt%ofdeanolandbetween24.5and27wt%of
SLS.Thismeansthatonlyforthoseonentrationsthe
miellesare intheorret rangeoflengthand rigidity
toinduetheappearaneoftheN
phasewithinrease
inSLSontent.
Inverted phase sequene I{H{N
our, however,
byinreasingdeanolontent,uponSLSonentration
largerthan26wt%. Suhinversionmustbedue,
there-fore, to a spei eet of deanol, whih possibly
af-fetsthegrowthandexibilityofthemixedmiellesin
anon-trivialway. Theinversionoursintheontextof
theompletesequeneI-H-N
-N
d
-L,whihappears
onlywithinreaseindeanolontent. Weshallnot
dis-uss now two small biaxial islands [29℄, not shown in
Fig.1beausetheydo notappearalong theA and B
lines,hereanalyzedin detail.
Abetterunderstandingofthedeanoleeton
mi-ellarstruture andphasetransitions was reently
ob-tainedthroughthestudyofthephasesequeneI-H
-N
byinvestigatingtheternarysystemvarying M
d for
xed values of the water: SLS molar ratio (M
w ). It
was shown [14,15℄ that the inverted sequene appears
already for M
w
= 45.2 (line A of gure 1). For suh
highwaterontenttheHdomainexistsonlyforavery
narrowinterval, M 0:195. So, the line A ontains
in fat theH phasein thesequene I-H-N
andpasses
verynearthetriplepoint. TheHphase'sfeaturesover
alargerrangeofM
d
values,byinvestigatingsamplesat
M
w
=39.4(lineBofFig. 2), was alsoperformed[16℄
followingthesequeneI-H-N
.
Theresultsdisussedintheprevioussetion3
on-rma\rossover"fromthenite/rigidmodel[21℄tothe
innite/exible model [22,23℄for theH phasewith an
inreasein thedeanolontent. Thiseetisprobably
due essentially to miellar growth with deanol
addi-tion. Suh growth ours in a slighter form
(anisome-tries inreasingfrom2.4to3) inI phase[30℄ butmust
begreatlyenhanedbyouplingwithorientationaland
positionalorderintheHphase[21-23℄.
Isotropic
a
0
10
20
30
0.0
0.5
1.0
b
c
d
e
f
Crystal
Columnar
Nematic
v
p
F
~T
Figure3. Theoretialphasediagramsforrods,takenfrom
[21℄. The omplex path proposed in this work (and
de-sribed in the text) orresponds to the urve drawn here
aroundthetriplepoint,allowingfortheobservedphase
se-queneI-H(olumnar)-N
.
Inthe binary system mielles are short in I phase
andlongerinHphase,growingonsiderablyintheI-H
oexistenerange [13,25℄. FromX-rayresults [13℄ the
estimated lengthin phaseHgivesL/Din therange
6-16. The behaviora 1=3
v
assures [9℄ that mielles
are in thenite rigidregime,and thereforemust have
alargepersistenelength,orPL.Nonematiphase
exists inthebinarysystem,so thesystemisabovethe
triplepointattheI-Htransition.
Thetheoriesofself-assemblythatpreditI-(N)-H
phasetransitions[21-23℄areneitherspeiforternary
systems,nordealwiththespeiroleofdeanol.
How-ever, the eet of deanol might be dedued through
omparison ofthe experimentalphase diagramof Fig.
1withthetheoretialpreditions,showninFig. 3. The
rstpointtoemphasizeisthatdeanolbringsI-H
withoutdeanol. InthebinarysystemI-Hdiret
tran-sitionours[25℄for40wt%ofSLS,whileitanour
for lessthan26wt%forabout3.5wt%ofdeanol(see
Fig. 1). This shows that thepartile volume fration
p
, whih denes the transition point, is neither
pro-portionalto theSLSnortothetotalamphiphile (SLS
+deanol)weightonentration,andmust greatly
in-reasewith deanoladdition.
Let us rst ompare the experimental phase
dia-gram with thephasediagram forrigid rod[21℄. From
the position of the triple point we onlude that the
deanol ontent plays the role of the assoiation free
energy . A slight derease in deanol (from 4 wt%
to 3.7 wt%)promotesahangefrom I-N-Hto I-H
be-havior. It shows that inreasein deanol should have
the sameeet of aninrease in theabsolute valueof
(oralternativelyadereaseintemperature). Thisis
onsistent with the eet of miellar growth with
de-anol additionmentionedbefore. Fromthetheoretial
phase diagramforrigid rodstheH-N line anonlybe
rossed throughaderease in partilevolumefration
p
. The H-N
transition is experimentally obtained
with inrease in
v and
vpar
(sine M
d
inreases at
onstant M
w
), but this does not mean neessarily an
inrease in
p
. With the onstant R
tot
value used in
Fig. 2,
p
showsa slight inreasewith M
d
. Butif we
allow for asmall derease in R
tot
, as indiatedin the
struturalanalysis thatgaveadereaseof4%inR
eff ,
p
an atually derease as M
d
inreases. It is thus
possiblethat,dueto aombinedeetof marked
par-tile growth and smallderease in polar head size,
p
dereases within the H domain, induing the H - N
transition. Thepartilevolumefrationinthe
hexago-nalplaneislearlymuhsmallerintheternarythanin
thebinarysystem,whilethefrationintheuid
dire-tion inreases in the ternary system. So, deanol has
theeetofinitiallyinrease
p
,duetomiellargrowth
and inreasein miellarlength,but inexessprodues
asmalldereasein
p
,due toitseetin dereasethe
miellediameter.
Toompletethepitureitisneessaryto takeinto
aount that thenearbynemati phase N
is made up
of small mielles[31℄,while miellesare longin theH
phase. Thisanberationalizedonlyadmittingthat
mi-ellesmustbeomeshorterattheH{N
transitionand,
onversely,thatalargeinreaseinmiellarlengthmust
ourattheN
-Htransition. Suhfatisqualitatively
understandable, sine miellar growth is triggered by
both orientationaland positional order [21-23℄. What
experimental results show is that, sine anisometries
are nearly equalin the I phase ( 3) before theI
-H transition [14,30℄ and in the N
phase after the H
- N
transition [31℄,growth ours muh moredue to
positionalordering thandue toorientationalordering.
Enhanement of growth in the H phase, asompared
to theNphase,arisestheoretially[23℄from detailsof
Thenon-trivialI-H-N
sequeneintheSLSternary
system with inrease in M
d
may be therefore
under-stood onbasisofaomplexpath throughthe
theoret-ialphase diagramforrigid rods[21℄. I- Htransition
oursabovethetriplepoint,forsmalllengthsandlarge
volume fration in I phase, with large inrease in
mi-ellarlength at I - H oexistene. Alongthe H phase
inreasein M
d
orrespondstofurther miellargrowth,
arriving to the \innite end-to-end" limit. This path
goes, therefore, around the triple point (whih
orre-spondsto3for
p
0:45),withaninreaseinsize
anddereasein
p . H-N
transitionours belowthe
triplepoint witha markedderease in miellarlength
attheH-N
oexistene.
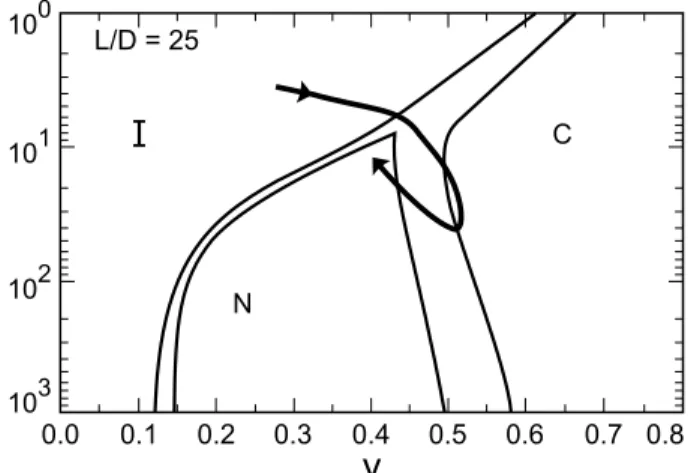
10
0
10
1
10
2
10
3
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.7 0.8
C
N
L/D = 25
v
Figure4. Theoretialphasediagramforexiblerodstaken
from[22℄. Ordenate P/D(seetext)inlog sale asa
fun-tionof volume fration V. Theomplex pathproposed in
this work (and desribed in the text) orresponds to the
urvedrawnherearound thetriple point, allowing for the
observedphasesequeneI-H(olumnar)-N.
Let us now ompare the experimental phase
dia-gramalsowiththepreditionsforexiblerods[22,23℄,
sine resultsshowed that the H phase is in the
\in-nite/exible" limit. It has been shown that the
exis-teneoftheNphaserequiresaertainrigidityof
semi-exiblemielles. ComparisonofthetriplepointofFig.
1 with the theoretial phase diagram (Fig. 4) shows
thatinreaseindeanolontentmustindueaninrease
intheP/Dratioofalongexiblerod. Thatmeansthat
inrease in deanol should inrease rod rigidity, and
not promote exibility. The mielles in the H phase
of the binary system are quite rigid, sine they have
rigid behavior and are long (L/D 10). Therefore
theyhaveP L. Inrease in rigidityanbeobtained
either by inrease in P/D or derease in L/D. Sine
deanolpromotesmiellergrowth(inreasein L/D),it
must promote also marked inrease in P/D in order
to allow the H - N
transition within the theoretial
framework of exible rods. Inrease in rigidity of rod
like mielleswith deanol addition anbe understood
anol (p
o
1), responsible for its bending energy at
the miellar polar / apolar interfae, is onsiderably
larger than that of SLS (p
o
1=3), and the
param-eter of the mixed region will be also orrespondingly
larger[24℄. Thus deanolpromotes rigidityin rod like
mielles, evenif may eventuallypromote exibility in
planar membranes, due to the planar symmetry
om-binedwithdereaseinhargeofpolarsurfaes.
Thediret I -H transition ours abovethetriple
point(whih oursforP/D13forlongexiblerods
[22,23℄). Inrease in M
d
orresponds again to a path
\turningaroundthetriplepoint",withinreaseinL/D
andP/D.ThetransitionlineH-N
isrossedalsowith
a derease in
p
, ombined with inrease in P/D. It
shouldberemarkedthat,inoneofthetheoretialphase
diagrams[22℄,itispossibleaslightlyre-entrant
behav-ior (for L/D 25), whih allows to ross the H - N
line atxed
p
, butwith averylargeinreasein P/D
(ofabout3ordersofmagnitude). Howeveraderease
in
p
seems moreplausible. Moreover, as the innite
limitisbeingapproahed,theoupanyinthe
hexag-onalplane, givenby2R =a, whih islearly dereasing
withM
d
, playstheroleof
p .
In onlusion, the experimental fats an be
ex-plained within the frameworkof the theoretial phase
diagrams for self-assembly systems, but with arather
omplexpathunifying bothphasediagrams. The
\in-nite/exible"limitbehaviorobservedfortheHphase
of the ternary system annot in fat be asribed
nei-therto \innite"nor\exible" mielles,sinedeanol
promotes growth, but not exibility. There must be
thereforealimiton thevalidityof. Eq. (3a), possibly
relatedto aminimumvalueof 2R/athat is onsistent
withpartitionofwaterinthethreedimensions. Bellow
suhlimit,Eq. (3b) holds. Thusthe\innite/exible"
limitisreahedin theternarysystembeausedeanol
is unable to form mielleson its own, beingfored to
enterontheSLSmielles,ausingmiellargrowthand
dereasein 2R =a. Thereal ternarysystemrossesthe
I{Hlinewithinthe\nite/hard"framework,then
mi-ellargrowthwithinreaseinM
d
induesapassageto
the\innite"framework,whihtogetherwithderease
in
p
indues nally the rossing of the H { N
line,
and this transition is again within the framework of
the\nite/hard"limit.
VI The N
(N
b
) N
d
phase
se-quene
This sequene has not been worked out yet
theoret-ially within the framework of self-assembly theories,
whih onsider the mielle symmetry as a priori
de-ned,andexistuptonowonlyfortheI{N
{HandI
{N
d
{Lsequenes,thatreferrespetivelytorodsand
diss. It isoutof thesopeofthepresentpaperto
re-donottakeself-assemblyintoaount,andalsodonot
aountfortheneighboringHandL phaseswith
posi-tionalorder,aswellastheextensiveworkdonebyother
groupsin theseondorderphasetransitionswith
tem-peraturethat ourin apartiularsystem(potassium
laurate/water/deanol).
However,theN
-N
d
transitionhasbeenshown
ex-perimentally [19,24℄ to be a funtion of deanol:SLS
molar ratio (M
d
) in systems with three dierent
am-phiphiles, ourring always around similar M
d
val-ues. Furthermore, it wasshown [32℄ that the system
SLS/water/deanolhasarstorderphaseN
-N
d
tran-sitionatsuhM
d
value. Twosmallbiaxialislandshave
been later disovered [29℄, evidening also rst order
transitions and even the existeneof two dierent
bi-axialphases(N
b+ andN
b
),stronglyindiating
miel-larshapetransformations. Clearly,N
-N
d
diret phase
transitions exist, and phase N
b
does not our
nees-sarilyinbetween.
Wehavedevelopedanelasti bending theoryfor a
\singlemielle",basedonthesurfatantparameterp
o ,
whihis ableto aountfor shapetransformations. It
has beenshown [28℄ that a form transformation from
prolate spheroids to spheroylinders (SC) ours, as
the miellar anisotropy inreases (for > 1:8), due
to the bending energy of the polar/apolar interfae.
Suh transformation is a pre-requisite for I - H
tran-sition, sinethe SC form allowspolydisperse grow. It
hasalsobeenshown[33℄thatitisenergetiallyfavored
inSCmixed miellesthatdeanolstaysinthebodyof
theylinder, leavingunmixed amphiphilein the
hemi-spherialaps. Thetendeny of deanolto loalizein
the body of SC mielles ours in order to derease
the eletrostatiinteration between amphiphile polar
heads, and is responsiblefor miellargrowth with
de-anol addition. The shorter mielles in the N
phase
will have a larger deanol mole fration in the body
of the SC (here named y) than in the H phase, but
still without deanol in the hemispherial aps. Only
withfurther deanolinrease,attheN
-N
d
transition,
the deanol of theylindrial body imposes amarked
hangeinmiellarform(fromSCtoplatelet),as
exten-sivelystudiedby Amaralet al[24℄. A synthesisof the
obtainedresultsisshownin table1. Theexperimental
M
d
valueand the known SC anisometry , at the N
{ N
d
transition, give an \experimental" y, whih an
be ompared to the preditionsof the elastibending
model.
Table1showsaonstanty valueforthethree
am-phiphiles. NotethattheM
w
valuesatthetransitionfor
the three amphiphiles are rather dierent[19℄, due to
dierent miellarsizes and waterbinding. The elasti
bendingtheory,forasinglemixedmielle,orretly
pre-dits[24℄atransformationatthisy valueforexpeted
p
o
valuesof amphiphile and osurfatant, without
This form transformation, besides oiniding with N
{N
d
transitions, oursalso intheI phase,asstudied
bydetailed analysis[17℄ofurvesofsmall angleX-ray
sattering intheSLS/water/deanolsystem.
TableI.Experimental M
d
values(deanol/amphiphile
moleular ratio), estimated anisometries of the SC
and relatedy values(deanol/amphiphileratiosin the
body of the SC), at the N
-N
d
transition, taken from
[24℄. SDS, KL and SLS are, respetively, the
am-phiphiles sodium deyl sulfate, potassiumlaurateand
sodiumdodeylsulfate.
SDS KL SLS
M
d
0.300.03 0.380.02 0.380.01
2.30.3 3.00.3 3.0 0.3
y 0.650.03 0.640.03 0.640.03
Thisideaof shapetransformationat theN
- (N
b )
-N
d
phasesequene inspiredaproposalofonsidering
thestatistialmehanisofpolydisperseuniaxialforms,
whih wasworkedoutbyHenriques &Henriques[34℄,
leading to aphase diagram with the typial topology
oftheI{Ntransitions,inludingthetwouniaxialand
onebiaxialphases. Phasetransitionswithtemperature
are inthis model orrelatedwithdierent populations
ofthetwouniaxialforms.
To sum up, the sequene I - H - N
- N
d
- L an
beunderstoodbya\superposition"oftheoriesof
self-assembly (whih onsider the mielle symmetry as a
prioridened)withanelastibendingtheoryfora
\sin-glemielle"(whihaountsforshapetransformation).
Itremainstobeworkedoutanextensionofsuhelasti
theoryto astatistialensembleofmielles.
Aknowledgments
Theauthorthanksthe ontributionofthe
ollabo-ratorsoftheworksherereviewed,partiularlyDrs. R.
Itri,P.Mariani,G.Taddei,O.SantinFilho,V.
Castel-letto and C. V. Teixeira, and thenanial support of
PRONEX/CNPq/MCTandFAPESP.
Referenes
[1℄ V. Luzzati, Biologial Membranes, ed. D.Chapman,
AademiPress,NewYork,pp.1-123(1968).
[2℄ P.Ekwall,AdvanesinLiquidCrystals,ed.G.H.Brown,
AademiPress,London,vol.1,pp.1-142(1975).
[3℄ J.N.Israelahvili,D.J.Mithell,andB.W.Ninham,J.
Chem.So.FaradayTrans.II72,1525 (1976).
[4℄ P.Mariani,V.Luzzati,andH.Delaroix,J.Mol.Biol.
204,165(1988).
[5℄ J.B. Forrest and L.W. Reeves, Chem. Rev. 81, 1
(1981).
[6℄ L.Q. Amaral,C.A. Pimentel, M.R. Tavares, andJ.A.
[7℄ J. Charvolin, A.M. Levelut, and E.T. Samulski, J.
PhysiqueLett.40,L-587(1979).
[8℄ L.J. Yu and A. Saupe, Phys. Rev. Lett. 45, 1000
(1980).
[9℄ L.Q.Amaral,A.Gulik, R.Itri,andP.Mariani. Phys.
Rev.A46,3548(1992).
[10℄ L.Q.Amaral, R. Itri, P. Mariani, and R. Miheletto,
Liq.Crystals12,913(1992).
[11℄ P. Mariani and L.Q. Amaral, Phys. Rev.E 50, 1678
(1994).
[12℄ P.Mariani,L.Q.Amaral,L.Saturni,andH.Delaroix,
J.Phys.II(Frane)4,1393(1994).
[13℄ R.Itri,L.Q.Amaral,andP.Mariani,Phys.Rev.E54,
5211(1996).
[14℄ R.ItriandL.Q.Amaral,Phys.Rev.E58,1173(1998).
[15℄ C.V.Teixeira,R.Itri,andL.Q.Amaral,Langmuir15,
936(1999).
[16℄ O. Santin Filho, R. Itri, and L.Q. Amaral, J. Phys.
Chem.104,959(2000).
[17℄ C.V.Teixeira,R.Itri,andL.Q.Amaral,Langmuir16,
6102(2000).
[18℄ L.Q. Amaral, M.E.M. Helene, D.R. Bittenourt, and
R.Itri,J.Phys.Chem.91,5949(1987).
[19℄ L.Q.AmaralandM.E.M.Helene, J.Phys.Chem.92,
6094(1988).
[20℄ P.O.Quist,B.Halle,andI.Furo,J.Chem.Phys.95,
6945(1991).
[21℄ M.P. Taylor and J. Herzfeld, Phys. Rev.A 43, 1892
(1991).
[22℄ R.Hentshkeand J.Herzfeld,Phys.Rev.A 44,1148
(1991).
[23℄ P.vanderShoot,J.Chem.Phys.104,1130(1996).
[24℄ L.Q.Amaral,O.SantinFilho,G.Taddei,andN.
Vila-Romeu,Langmuir13,5016(1997).
[25℄ R. Itri and L.Q. Amaral, J. Phys. Chem. 94, 2198
(1990).
[26℄ V. Castelletto, R. Itri, and L.Q. Amaral, J. Chem.
Phys.107,638(1997).
[27℄ V.CastellettoandL.Q.Amaral,J.Phys.Chem.B103,
8877(1999).
[28℄ G.TaddeiandL.Q.Amaral,J.Phys.Chem.96,6102
(1992).
[29℄ P.O.Quist,Liq.Cryst.18,623(1995).
[30℄ R.ItriandL.Q.Amaral,Phys.Rev.E47,2551(1993).
[31℄ P.O.Quist,B.Halle,andI.Furo,J.Chem.Phys.96,
3875(1992).
[32℄ L.Q.Amaral,LiquidCrystals7,877(1990).
[33℄ W.M. Gelbart, W.E. MMullen, A. Master, and A.
Ben-Shaul,Langmuir1,101(1985).
[34℄ E.F. Henriques and V.B. Henriques, J. Chem. Phys.